Remember that carboncarbon single bonds can rotate, whereas carboncarbon double bonds cannot rotate. How many different structures
Question:
Remember that carbon–carbon single bonds can rotate, whereas carbon–carbon double bonds cannot rotate. How many different structures are shown below?
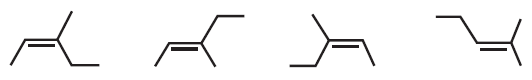
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
The first and the t...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
Many naturally occurring substances contain several carbon-carbon double bonds: some isolated, some conjugated, and some cumulated. Identify the types of carbon-carbon double bonds found in each of...
-
(a) Calculate the energy needed to compress three carbon-carbon single bonds and stretch three carbon-carbon double bonds to the benzene bond length 1.397 . Assume a harmonicoscillator...
-
How might Kekule explain the fact that there is only one dibromobenzene with the bromines on adjacent carbon atoms, even though we can draw two different structures, with either a double or a single...
-
Partners have fiduciary obligations to each other. Explain what this means and give examples.
-
On July 1, 2014, Katrina purchased tax-exempt bonds (face value of $75,000) for $82,000. The bonds mature in five years, and the annual interest rate is 6%. The market rate of interest is 2%. a. How...
-
Find sin 120 by using 120 = 90 + 30.
-
Sandra and Thomas McGuire entered into a purchase-and-sale agreement for Beccas Boutique with Pascal and Rebecca Tursi. The agreement provided that the McGuires would buy the store for \($75,000,\)...
-
Adria Lopez is considering the purchase of equipment for Success Systems that would allow the company to add a new product to its computer furniture line. The equipment is expected to cost $ 300,000...
-
Appendix A UX and CX - for coffee and tea drinkers Find a way of making a tea and coffee machines more interesting. Rather than just having a coffee machine which will make a cappuccino, tea or latte...
-
Lillian purchased a guitar from Smash Music Stores. It regularly sold for $670, but was on sale at 10% off. She paid 8% tax. She bought it on the installment plan and paid 15% of the total cost with...
-
What are the chemical formulas for the following structures? (b) (a) (c) (d)
-
Which of the structures shown in the previous question are either the cis- or the trans-isomers of 3-methyl-2-pentene? Circle them.
-
The number of hours per week of television viewing for random samples of fifth grade boys and fifth grade girls were obtained. Each student logged his or her hours for one Monday-through-Friday...
-
What is the direct source of energy for the ATP produced by ATP synthase?
-
If unions are to be successful in obtaining wage rate increases for their members,they must be able to cause a shift in either the labour demand curve or the labour supply curve or both.? Explain how...
-
Solving Worldly Issues with Entrepreneurship For this project, you will apply your entrepreneurial and enterprising skills to a global issue or problem. Use the following steps to structure your...
-
Who develops Kali Linux? What distribution was the predecessor to Kali Linux? 2. What is the main purpose(s) of the Kali Linux distribution? 3. What are the installation options for Kali Linux?...
-
Have you heard of "The Five Forces model" popularized by Harvard professor Michael Porter, that is a framework for analyzing the attractiveness of an industry? These readings came from the book...
-
With U.S. Census Bureau data, the model that relates the average annual earnings (in thousands of dollars) of females, F, and males, M, with various levels of educational attainment was found to be F...
-
Find the cross product a x b and verify that it is orthogonal to both a and b. a = (t, 1, 1/t), b = (t 2 , t 2 , 1)
-
A 500 g particle moving along the x-axis experiences the force shown in FIGURE EX9.21. The particles velocity is 2.0 m/s at x = 0 m. What is its velocity at x = 3 m? F; (N) 15 - 10- 5- -x (m) 0- 1...
-
A 2.0 kg particle moving along the x-axis experiences the force shown in FIGURE EX9.22. The particles velocity is 4.0 m/s at x = 0 m. What is its velocity at x = 2 m and x = 4 m? F; (N) 10- -x (m) 4...
-
A particle moving on the x-axis experiences a force given by F x = qx 2 , where q is a constant. How much work is done on the particle as it moves from x = 0 to x = d?
-
In Boston, due to the large amount of snowfall during winter of 2014-2015, the city has brought in several "snow dragons" to melt the snow. Model SND5400 is rated at: 180 ton snow rated capacity per...
-
How does the three-tier model effectively meet the literacy development needs of individual students? How does the response to intervention model integrate the three-tiers of instruction? What are...
-
TCS was established in 1968 with its headquarters in Mumbai. It was formed as a division of Tata Sons Limited (TSL), one of India's largest business conglomerates, and was called 'Tata Computer...

Study smarter with the SolutionInn App


