Estimate the flow rate (kg/min and kmol/min) of each component and the composition (mole fractions) of the
Question:
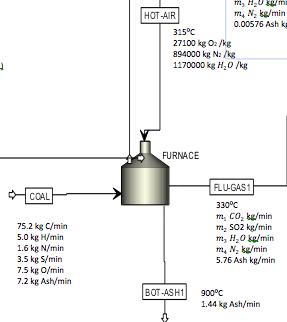
Estimate the flow rate (kg/min and kmol/min) of each component and the composition (mole fractions) of the furnace flue gas. (Ignore the fly ash in calculating mole fractions.) At what rate (kg/min) is fly ash removed from the flue gas by the electrostatic precipitator?
Transcribed Image Text:
HOT-AIR m, N, kg/min 0.00576 Ash ka 315°C 27100 kg O: /kg 894000 kg N: /kg 1170000 kg H,0 /kg FURNACE FLU-GAS1 COAL 330°C 75.2 kg C/min 5.0 kg H/min 1.6 kg N/min 3.5 kg S/min 7.5 kg O/min 7.2 kg Ash/min m, CO, ke/min m, 502 kg/min m, H,0 ku/min m, N, kg/min 5.76 Ash kg/min BOT-ASH1 900°C 1.44 kg Ash/min
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Flow rate kgmin CO2 752 kg Cmin 50 kg Hmin 16 kg Nmin 35 kg Smin 75 kg Omin 72 kg Ashmin 1004 kgmin ...View the full answer

Answered By

SHINKI JALHOTRA
I have worked with other sites like Course Hero as a tutor and I have great knowledge on IT skills.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Question Posted:
Students also viewed these Business questions
-
Estimate the molar flow rate (kmol/min) of each element in the coal (other than those in the ash).
-
If 15% excess O 2 is fed to the combustion furnace, estimate the following: a. The oxygen and nitrogen feed rates (kmol/min). b. The mole fraction of water in the wet air, the average molecular...
-
The system may be assumed to meet the standard of 90% removal of the SO 2 released upon combustion. a. Determine the flow rate (kg/min and kmol/min) of each component in the cleaned flue gas leaving...
-
Seeking for information of a specific organization then answer following questions: (1) Give a brief introduction of the chosen organization. (2) Explain the type of the chosen organization. What are...
-
Of the seven three-dimensional primitive lattices, which ones have a unit cell where no two lattice vectors are perpendicular to each other?
-
Youve just joined the investment banking firm of Dewey, Cheatum, and Howe. Theyve offered you two different salary arrangements. You can have $90,000 per year for the next two years, or you can have...
-
Suppose that we have two independent samples, say Two models can be fit to these samples, \[\begin{gathered}y_{i}=\beta_{0}+\beta_{1} x_{i}+\varepsilon_{i}, \quad i=1,2, \ldots, n_{2}...
-
Limestone (calcium carbonate) particles are stored in 50-L bags. The void fraction of the particulate matter is 0.30 (liter of void space per liter of total volume) and the specific gravity of solid...
-
5. Add a worksheet labeled 'Pivot Chart' and move it so that it's the first one on the left. Then create a Pivot Chart (Column Chart) showing Country Name and Sum of USD Total. Format all cells...
-
A vertical aerial photograph reveals a tall building. The foot of one corner of the building has (x, y) coordinates (30.5, 62.0) (both measured in mm from the lower left-hand corner of the negative),...
-
Determine the feed rate (kmol/min) of O 2 required for complete combustion of the coal.
-
At what rate is heat removed from the furnace? Assuming that all of the heat removed from the furnace is used to generate steam (i.e., none is lost to the surroundings), estimate the rate of steam...
-
True or false: A logic pulser can force a node LOW or HIGH for extended periods of time.
-
Explain why managers need not diversify the firms they run.
-
How do bankruptcy and agency costs alter the effect of financial leverage on firm value?
-
How are the gains from leverage affected by a difference in the personal tax rates on stock versus interest income?
-
What is the argument behind the prediction that dividend payments and share repurchases should not affect shareholder wealth? What does the evidence say about how stock prices react to shareholder...
-
A firm with no debt expects to generate pretax net operating profit of $100,000 per year in perpetuity. The required return on assets for this firm is 10%, and it faces a corporate tax of 21%. a....
-
A company believes that there might be substantial doubt that it can meet its financial obligations as they come due for one year from the issuance of its financial statements. What plans might...
-
Horse serum containing specific antibody to snake venom has been a successful approach to treating snakebite in humans. How do you think this anti-venom could be generated? What are some advantages...
-
Convert the following to appropriate ips units: (a) A length of 1.5 m. (b) A stress of 600 MPa. (c) A pressure of 160 kPa. (d) A section modulus of 1.84 (105) mm3. (e) A unit weight of 38.1 N/m. ( f...
-
Generally, final design results are rounded to or fixed to three digits because the given data cannot justify a greater display. In addition, prefixes should be selected so as to limit number strings...
-
Repeat Prob. 111 for: (a) = F/A, where A = d2/4, F = 120 kN, and d = 20 mm. (b) = 32 Fa/d3, where F = 800 N, a = 800 mm, and d = 32 mm. (c) Z = (/32d)(d 4 d 4 ) for d = 36 mm and d i = 26 mm. (d)...
-
York's outstanding stock consists of 80,000 shares of cumulative 7.5% preferred stock with a $5 par value and also 200,000 shares of common stock with a $1 par value. During its first four years of...
-
Hudson Community College enrolls students in two departments, Liberal Arts and Sciences. The college also has two service departments, the Library and the Computing Services Department. The usage of...
-
Robert is designing a new Active Directory Domain Services infrastructure for a company called Litware, Inc., which has its headquarters in New York and two additional offices in London and Tokyo....

Study smarter with the SolutionInn App


