Both pipes in the system shown have an equivalent sand roughness k s of 0.10 mm and
Question:
Both pipes in the system shown have an equivalent sand roughness ks of 0.10 mm and a flow rate of 0.1 m3/s, with D1 = 12 cm, L1 = 60 m, D2 = 24 cm, and L2 = 120 m. Determine the difference in the water-surface elevation between the two reservoirs.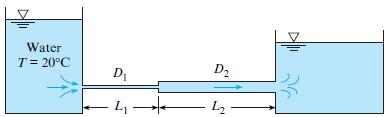
Transcribed Image Text:
Water T= 20°C D1 D2 - 4 L2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Equivalent Roughness k 010 mm Flow rate Q 01msec Diameter of Pipe 1 D 12cm 012m Length of Pipe 1 L 6...View the full answer

Answered By

NITHIN SHAJEENDRAN
I worked in chegg solving problems
0.00
0 Reviews
10+ Question Solved
Related Book For 

Engineering Fluid Mechanics
ISBN: 9781118880685
11th Edition
Authors: Donald F. Elger, Barbara A. LeBret, Clayton T. Crowe, John A. Robertson
Question Posted:
Students also viewed these Engineering questions
-
A linear actuator is used in the system shown in Figure P8.10 to position a mass M. The actual position of the mass is measured by a slide wire resistor, and thus H{s) = 1.0. The amplifier gain is...
-
In the system shown in Fig. 5.54, block A has mass mA, block B has mass mB' and the rope connecting them has a nonzero mass mole. The rope has a total length L, and the pulley has a very small...
-
In the system shown three masses are connected via string and a series of pulleys to one another. Find the acceleration of all blocks. Do not assume the slope to be fictionless, but assume that block...
-
Pelicans tuck their wings and free fall straight down when diving for fish. Suppose a pelican starts it dive from a height of 16.0m and cannot change its path once committed. If it takes a fish 0.20s...
-
Explain the four different types of unemployment. Briefly provide ways in which each type of unemployment might be reduced.
-
The equilibrium constant K c for the reaction N 2 (g) + O 2 (g) 2 NO(g) at 1200C is 1.00 * 10 5 . Calculate the equilibrium molar concentrations of NO, N 2 , and O 2 in a reaction vessel of volume...
-
An assembly line has a staple gun that rolls to the left at \(1.0 \mathrm{~m} / \mathrm{s}\) while parts to be stapled roll past it to the right at \(3.0 \mathrm{~m} / \mathrm{s}\). The staple gun...
-
Aaron Rivers, CPA, is auditing the financial statements of Charger Company, a client for the past five years. During past audits of Charger, Rivers identified some immaterial misstatements (most of...
-
Provide the products in each of the following reactions. Indicate the formation of any chiral carbons, and whether the product is or is not optically active. HINT: You may find it helpful to review...
-
Selected comparative financial statement information of Bluegrass Corporation follows. Required 1. Compute each years current ratio. Round ratios to one decimal. 2. Express the income statement data...
-
Water is pumped at a rate of 32 m 3 /s from the reservoir and out through the pipe, which has a diameter of 1.50 m. What power must be supplied to the water to effect this discharge? Elevation = 140...
-
Liquid discharges from a tank through the piping system shown. There is a venturi section at A and a sudden contraction at B. The liquid discharges to the atmosphere. Sketch the energy and hydraulic...
-
For a circuit with R = 1.00 k, L = 50.0 mH, and f = 10.0 kHz: (a) Find the impedance (in ohms), (b) Find the phase angle, (c) Find the current if the voltage is 15.0 V.
-
Rubina Care Ltd (Rubina) operates several nursing homes in South Australia. All residents are changed a fixed fee per week as set by legislation. Due to the quality of the care provided, occupancy...
-
As the newest BFM in the program office, you notice a disbursement for the X-86 Aircraft landing tail hook in the accounting system. You then notice that the disbursement is not matched to an...
-
Estimate year one proforma effective gross income (egi) based on the following assumptions: 1) for an apartment property with 250 - 2 bed-2 bath units property had an egi of $3,350,000 in the past 12...
-
A California couple, age 65 years each, retired to Arizona. They sold their California house for $500,000, realizing a $125,000 capital gain. They purchased a home in Arizona in the same year for...
-
Reflect on The Scream' by Kent Monkman: What might lawful Indigenous child apprehension teach us about the rule of order in Canada?
-
Suppose that you have a glass tube filled with atomic hydrogen gas (H, not H2). Assume that the atoms start out in their ground states. You illuminate the gas with monochromatic light of various...
-
$10,000 was borrowed at 3.5% on July 17. The borrower repaid $5000 on August 12, and $2000 on September 18. What final payment is required on November 12 to fully repay the loan?
-
A refrigerator uses refrigerant-134a as the working fluid and operates on the vapor-compression refrigeration cycle. The evaporator and condenser pressures are 200 kPa and 1400 kPa, respectively. The...
-
Repeat Prob. 1119E using appropriate software if ammonia is used in place of refrigerant-134a. Data From Q#19: A refrigerator uses refrigerant-134a as its working fluid and operates on the ideal...
-
A refrigerator uses refrigerant-134a as its working fluid and operates on the ideal vapor-compression refrigeration cycle. The refrigerant evaporates at 5F and condenses at 180 psia. This unit serves...
-
USE THE FOLLOWING TO ANSWERS QUESTIONS 13 - 18. Rowan Manufacturing, Inc. and Martin Machinery Company are two small metal fabrication companies serving the aerospace industry. On June 30, 2020,...
-
USE THE FOLLOWING TO ANSWER QUESTIONS 1-6. At the beginning of 2020, Red Corporation purchased a 30% interest in Black Company for $125,000. On the acquisition date, Black's stockholders' equity was...
-
#10. Bay Pine Medical Center estimates that a capitated population of 50,000 would have the following base case utilization and total cost characteristics: In addition to medical costs, Bay Pines...

Study smarter with the SolutionInn App


