Find the loss coefficient K v of the partially closed valve that is required to reduce the
Question:
Find the loss coefficient Kv of the partially closed valve that is required to reduce the discharge to 50% of the flow with the valve wide-open as shown.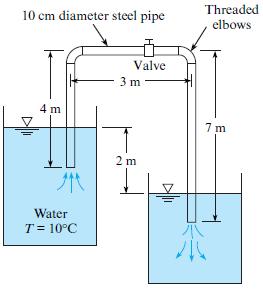
Transcribed Image Text:
Threaded elbows 10 cm diameter steel pipe Valve 3 m 4 m 7 m 2 m Water T = 10°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Given data Diametre of pipe D 10 cm Elevation differenceAz 2m Length ...View the full answer

Answered By

NITHIN SHAJEENDRAN
I worked in chegg solving problems
0.00
0 Reviews
10+ Question Solved
Related Book For 

Engineering Fluid Mechanics
ISBN: 9781118880685
11th Edition
Authors: Donald F. Elger, Barbara A. LeBret, Clayton T. Crowe, John A. Robertson
Question Posted:
Students also viewed these Engineering questions
-
What cathode potential (versus S.H.E.) is required to reduce 99.99% of Cd(II) from a solution containing 0.10M Cd(II) in 1.0 M ammonia if there is negligible current? Consider the following reactions...
-
Directions:For each question show all work that is required to arrive at the solution. Save this document with your answers and submit as an attachment to be graded. Use the triangle at the right....
-
Determine the 2 percentile that is required to construct each of the following CIs: (a) Confidence level = 95%, degrees of freedom = 24, one-sided (upper) (b) Confidence level = 99%, degrees of...
-
A particle of mass m moves in a certain plane P due to a force F whose magnitude is constant and whose vector rotates in that plane with a constant angular velocity . Assuming the particle to be...
-
The following data are for Argentina from 1985 to 2005. Year GDP Price Index 1985............................. 0.003 1986............................. 0.005 1991............................. 86.129...
-
(a) In an experiment, 5.0 mmol Cl 2 (g) was sealed into a reaction vessel of volume 2.0 L and heated to 1200. K, and the dissociation equilibrium was established. What is the equilibrium composition...
-
Peggy, standing on a moving railroad car, passes Ryan at velocity v. Peggy and Ryan both measure the length of the car, from one end to the other. The length Peggy measures is ___ the length Ryan...
-
What is meant by the term self-supporting growth rate? How is this rate related to the AFN equation, and how can that equation be used to calculate the self-supporting growth rate?
-
In what ways do declarative constraints in relational databases, such as PostgreSQL, contribute to data integrity and consistency enforcement, and how do they differ from imperative constraints ?
-
XYZ Ltd has three employees who are entitled to long service leave (LSL). The LSL can be taken after 15 years of service, at which time the employee is entitled to 11 weeks' leave. Information about...
-
Gasoline (T = 50F) is pumped from the gas tank of an automobile to the carburetor through a 1/4-in. fuel line of drawn tubing 10 ft long. The line has five 90 smooth bends with an r/d of 6. The...
-
The 12 cm galvanized steel pipe shown is 800 m long and discharges water into the atmosphere. The pipeline has an open globe valve and four threaded elbows; h 1 = 3 m and h 2 = 15 m. What is the...
-
Suppose X N(, ). a. Show via integration that E(X) = . b. Show via integration that V(X) = 2 .
-
Find the cardinal number of the indicated set. If n(A) = 40, n(B)=117, and n(AUB)=137, what is n(AB)?
-
Examine the intricate molecular machinery underlying the process of DNA replication in eukaryotic cells, elucidating the coordination of DNA polymerases, helicases, and DNA repair mechanisms to...
-
Assuming the sales price for Kayak was $306 per unit (instead of $244), how would the company allocate the 12,000 available machine hours between the two products so that it maximize its profits.
-
A rigid container and a piston contains 1L of R-134a at 100 o C and 3.6 MPa. In an isobaric quasi-equilibrium process, the gas expands to 1.2L. The control volume has a uniform cross section 0.5 m2...
-
Waylon's mother has just received notification that he will be fully reevaluated for special education services. This is the first time that Waylon has been fully reevaluated since qualifying for...
-
Explain why a population inversion is necessary in a laser.
-
If a force of F = 50 Ib is applied to the pads at A and C, determine the smallest dimension d required for equilibrium if the spring has an unstretched length of 1 ft. B 1 ft 1 ft F k = 15016/fr 1ft...
-
Repeat Prob. 1163E if the 30 psia evaporator is to be replaced with a 60 psia evaporator to serve a 15,000 Btu/h cooling load. Data From Q#63: A two-evaporator compression refrigeration system like...
-
A two-stage compression refrigeration system with an adiabatic liquid-vapor separation unit like that in Fig. P11 60 uses refrigerant-134a as the working fluid. The system operates the evaporator at...
-
A two-stage compression refrigeration system with an adiabatic liquid-vapor separation unit as shown in Fig. P1160 uses refrigerant-134a as the working fluid. The system operates the evaporator at...
-
On April 15, 2024, Cheyenne Ltd. declared a 50 per share cash dividend, payable on May 31 to shareholders of record on May 1. On the date of declaration, Cheyenne Ltd. had 264,000 common shares...
-
Harold Wright opened a medical practice in Santa Barbara, California, and had the following transactions during the month of January. Jan. 1 The business received $34,000 cash and gave capital to...
-
Management at Thomason Media has decided to allocate the costs of the paper's two support departments (administration and human resources) to the two support departments (advertising and...

Study smarter with the SolutionInn App


