Today, uranium-235 comprises only 0.72% of natural uranium; essentially all the rest is U-238. Use the half-lives
Question:
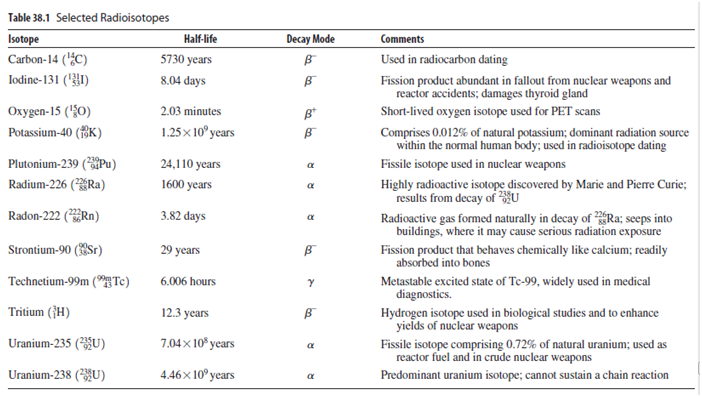
Today, uranium-235 comprises only 0.72% of natural uranium; essentially all the rest is U-238. Use the half-lives in Table 38.1 to determine the percentage of uranium-235 when Earth formed about 4.5 billion years ago.
Transcribed Image Text:
Table 38.1 Selected Radioisotopes Decay Mode Isotope Carbon-14 (¿C) Half-life Comments 5730 years Used in radiocarbon dating Iodine-131 ('1) 8.04 days Fission product abundant in fallout from nuclear weapons and reactor accidents; damages thyroid gland Охудеn-15 ({0) 2.03 minutes B* Short-lived oxygen isotope used for PET scans 1.25x 10° years Comprises 0.012% of natural potassium; dominant radiation source within the normal human body; used in radioisotope dating Potassium-40 (fK) Plutonium-239 (Pu) 24,110 years Fissile isotope used in nuclear weapons a Radium-226 (*ÝRa) 1600 years Highly radioactive isotope discovered by Marie and Pierre Curie; results from decay of HU Radioactive gas formed naturally in decay of Ra; seeps into buildings, where it may cause serious radiation exposure Fission product that behaves chemically like calcium; readily Radon-222 (Rn) 3.82 days Strontium-90 (Sr) 29 years absorbed into bones Technetium-99m ("HTc) 6.006 hours Metastable excited state of Te-99, widely used in medical diagnostics. Hydrogen isotope used in biological studies and to enhance yields of nuclear weapons Fissile isotope comprising 0.72% of natural uranium; used as reactor fuel and in crude nuclear weapons Tritium (}H) 12.3 years Uranium-235 (U) 7.04× 10* years Uranium-238 (U) 4.46× 10º years Predominant uranium isotope; cannot sustain a chain reaction
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
According to the NEA identified uranium resources total 55 ...View the full answer

Answered By

Dudhat Vaidehi
I tutored mostly elementary school students privately after school and during the summer. We met in their homes or at the public library. I charged an hourly fee, and I provided any necessary materials.
Having taught in special education in two local schools for many years meant that I had contact with a lot of parents of special needs students. I never had to advertise — word of mouth was how most folks knew of me. At one point I did have a website, but didn't utilize it much. I stayed very busy, especially in the summers, and always had a full schedule. I typically met with each student's teacher in order to get an idea of what the focus of my instruction/remediation should be. Becoming familiar with the student's learning style(s) was also very helpful. Often parents would share records and test results with me. After each tutoring session, I documented the student’s progress and gave parents written updates, as well as phone calls or emails as needed.
While my students and I certainly utilized technology and the internet often during our sessions, I never tutored online or for any tutoring company, so am not familiar with the curriculums or methods used in those settings.
Tutoring one on one was very enjoyable and rewarding. My students and I had fun, and grew quite fond of one another. The extra income was a bonus. I had to retire from tutoring due to a physically handicapping disease, and miss my students very much.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Naturally occurring uranium is composed mostly of 238U and 235U, with relative abundances of 99.28% and 0.72%, respectively. The half-life for 238U is 4.5 X 109 years, and the half-life for is 7.1 X...
-
In 1972, a worker at a nuclear fuel plant in France found that uranium from a mine in Oklo, in the African Republic of Gabon, had less U-235 than the normal 0.7% a quantity known from meteorites and...
-
Almost all of naturally occurring uranium is 238/92U with a half-life of 4.468 x 109 yr. Most of the rest of natural uranium is 235/92U with a half-life of 7.038 x 108yr. Today a sample contains...
-
4. Papo and Pepe are two barbers from a small barbershop. Theyhave their two court chairs plus two waiting chairs. The followingresults were found: P0 = 1/16 P1 = 4/16 P2 = 6/16 P3 = 4/16 a. What is...
-
Draw the structures of the following compounds. (Some parts may have more than one correct answer.) (a) An achiral trimethylcyclohexane with two chair forms that are conformational diastereomers. (b)...
-
Form the equal-weighted portfolio of the stocks in problem 1. (a) Compute i , 1 and i , 2 for the portfolio. (b) Compute the expected return of the portfolio. (c) Compute the portfolios risk...
-
Choose a country from three of the regions presented in Table 6.7. Using the Internet, collect as much information as you believe is needed to identify the potential for market segments based on age,...
-
The number of cars arriving per hour at Lundbergs Car Wash during the past 200 hours of operation is observed to be the following: NUMBER OF CARS ARRIVING FREQUENCY 3 or fewer ............. 0 4...
-
Discuss each of the following terms: (a) data (b) database (c) database management system (d) database application program (e) data independence (f) security (g) integrity (h) views 2. What is...
-
A leveraged ETF is an exchange-traded fund that uses debtor derivatives as leverage to amplify the returns of a benchmark index. Leveraged ETFs can produce significant short-term gains/losses, such...
-
Brachytherapy is a cancer treatment involving implantation of radioactive seeds at the tumor site. Iridium-192, often used for cancers of the head and neck, undergoes beta decay by electron capture...
-
Youre a geologist assessing underground sites for nuclear waste storage. A ruling by the U.S. Environmental Protection Agency suggests that waste-storage facilities should be designed for a million...
-
Pletcher Dental Clinic is a medium-sized dental service specializing in family dental care. The clinic is currently preparing the master budget for the first 2 quarters of 2020. All that remains in...
-
7) If the ratio of two-wheeler vehicle to the three-wheeler vehicles in a city is 12:15. Find the total number of three- wheeler vehicles if the total number of two-wheeler vehicles is 300.
-
Let U={12, 13, 14, 15, 16, 17, 18, 19, 20, 21} A={15, 16, 17, 18} B={12, 14, 16, 18, 20} C={13, 15, 16, 20, 21} Find the set. (AUB)' n C = 1 {} 0.0.... X 3 {}
-
web cache in the lower network. . R HTTP servers .50 .60 .30 .10 local webcache Suppose that: The capacity of the link connecting the upper and lower networks is R = 100Mbps (100 x 10 bits per...
-
Please use the readings you select to address the following question: why do people use drugs and what are the effects of drug use on individuals and their communities? Think of how the readings...
-
The following creditors have proved claims against Sarah's insolvent estate: SARS a claim of R120 000 (One hundred and twenty thousand rand) for income tax before sequestration; Best Funerals a claim...
-
The challenge with the Korean Money Market Hedge is that it is a payable - a payable form a Korean won cash balance. A MM Hedge for a payable is to simply transfer money into the target currency at...
-
H.J. Heinzs annual dividends were as follows: 1990 ..............$0.540 1991.............. 0.620 1992 .............. 0.700 1993.............. 0.780 1994 .............. 0.860 1995 .............. 0.940...
-
The post is made from 606l-T6 aluminum and has a diameter of 50 mm. It is fixed supported at A and B, and at its center C there is a coiled spring attached to the rigid collar. If the spring is...
-
The post is made from 606l-T6 aluminum and has a diameter of 50 mm. It is fixed supported at A and B, and at its center C there is a coiled spring attached to the rigid collar. If the spring is...
-
The A-36 steel wires AB and AD each have a diameter of 2 mm and the unloaded lengths of each wire are LAC = 1.60 m and LAB = LAD = 2.00 m. Determine the required diameter of wire AC so that each wire...
-
An asset pays $ 2 0 0 annually forever starting next year ( t = 1 ) . The discount rate for the asset is 6 . 7 % . What is the value today ( t = 0 ) of the asset?
-
A company's bond has an annual coupon rate of 4 % . The bond pays coupons semiannually and has a par value of $ 1 , 0 0 0 . The bond matures in 6 years. What is the percentage ( % ) yield to maturity...
-
A bond pays a semi-annual coupon at an APR of 11.25%. The bond will mature in 6.00 years and has a face value of $1,000.00. The bond has a yield-to-maturity of 11.82% APR. What is the current yield...

Study smarter with the SolutionInn App


