Question:
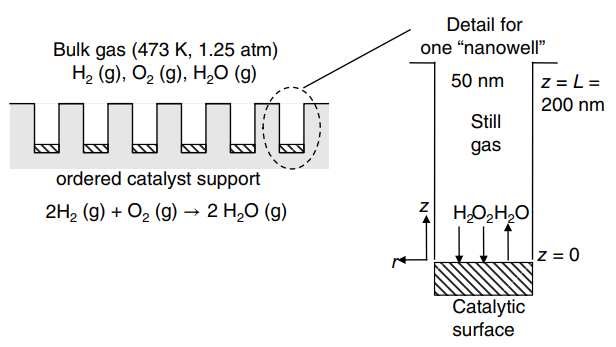
Consider the novel €œnanostructured€ catalyst surface shown in the figure below. The catalyst support consists of an ordered array of cylindrical nanowells€ of 50 nm diameter and 200 nm depth (1 nm = 10
-9m). A catalytic surface coats the bottom of each well. Although gas flows over the catalyst surface, the gas space within each €œwell€ is stagnant€”i.e., it is not well mixed. In the present application, the catalyst surface is used to convert unreacted H
2 gas (species A) and O
2gas (species B) from a fuel cell into H
2O vapor (species C according the reaction 2 H
2(g) + O
2(g) †’ 2H
2O(g). The reaction is con sidered diffusion limited within the catalyst well. The process is isothermal at 473 K and isobaric at 1.25 atm total system pressure, with bulk gas mole fraction compositions of Y
A,ˆž= 0.01, y
B,ˆž= O
2is by far the dominant species. What is the flux of H
2into the process under these conditions?
Transcribed Image Text:
Detail for one “nanowell" Bulk gas (473 K, 1.25 atm) На (9), Ог (9), Н,0 (9) 50 nm Z = L = 200 nm Still gas ordered catalyst support 2H2 (g) + O2 (g) → 2 H,0 (g) Н.О-Н,о Catalytic surface








