The table below presents equilibrium distribution data for four gaseous solutes dissolved in water, using air as
Question:
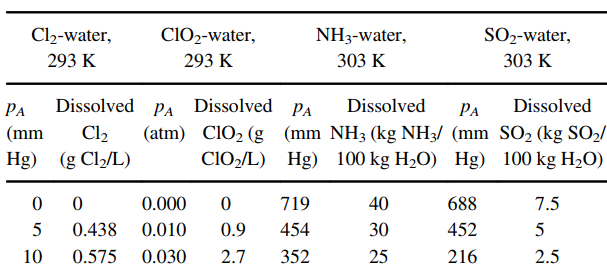
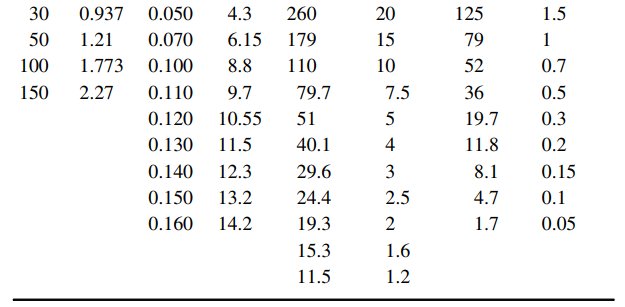
a. Using a spreadsheet to perform the calculations, prepare a graph of the equilibrium distribution data for each solute as partial pressure in the gas vs. molar concentration dissolved in the liquid (pA €“ cAL), and also in mole fraction coordinates (yA €“ xA) at 1.0 atm total system pressure. Which solute is the most soluble in water? Which solute dissolved in water can be stripped into air the easiest?
b. For each solute at the appropriate concentration range, estimate the Henry€™s law constant (H) based on the definition pA = H c*AL, and the distribution coefficient m based on the definition yA, = mx*A at 1.0 atm total system pressure.
DistributionThe word "distribution" has several meanings in the financial world, most of them pertaining to the payment of assets from a fund, account, or individual security to an investor or beneficiary. Retirement account distributions are among the most...
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Fundamentals Of Momentum Heat And Mass Transfer
ISBN: 9781118947463
6th Edition
Authors: James Welty, Gregory L. Rorrer, David G. Foster
Question Posted:





