The equilibrium constant for formation of CuCl 2 - is given by What is the solubility of
Question:
The equilibrium constant for formation of CuCl2- is given by
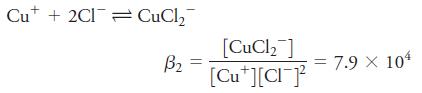
What is the solubility of CuCl in solutions having the following analytical NaCl concentrations:
(a) 5.0 M?
(b) 5.0 3 10-1 M?
(c) 5.0 3 10-2 M?
(d) 5.0 3 10-3 M?
(e) 5.0 3 10-4 M?
Transcribed Image Text:
Cu* + 2Cl= CuCl, [CuCl, ] B2 [Cu*][CI} = 7.9 X 104
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Write equation for complex formation of CuCl 2 where 4 is the complex formation constant for CuCl 2 Cu 3 2Cl CuCl 2 2 CuCl 2 Cu 2 Cl 2 79 10 4 CuCl 2 ...View the full answer

Answered By

Umair Expert
Hi Everyone.
I have 6 years of teaching experience.
I am serving as a tutor 2 more websites.
I will provide you projects and solutions of questions related to any subject.
I am a good programmer.(PHP, Python)
I am good command in mathematics.
I am here to assist you.
I'll be very happy to work with you...
Thanks dear..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
The equilibrium constant for the reaction H2 + at 1 atm and 1500C is given to be K. Of the reactions given below, all at 1500C, the reaction that has a different equilibrium constant is (a) H2 + 12O2...
-
The equilibrium constant for the H2 + ½ O2 H2O reaction at 1 atm and 1200 K is KP. Use this information to determine the equilibrium constant for the following reactions: (a) at l atm H, +...
-
The equilibrium constant for the reaction SO3 SO2 + O has the following values: Determine the average heat of dissociation using graphical method. T 800 K 900 K 1000 K 1105K | 0.0319 | 0.153 | 0.540...
-
Hrishi is a senior executive for a large manufacturing company in Mississauga Ontario where he has been employed for the past 10 years and his annual salary is $250,000 including bonus. He is 47...
-
Manuel bought a $100,000 bond with a 4% coupon for $92,300 when it had five years remaining to maturity. What was the prevailing market rate at the time Manuel purchased the bond? (Taken from CIFP...
-
Ceda Co. has equipment that cost $80,000 and that has been depreciated $50,000. Instructions Record the disposal under the following assumptions. (a) It was scrapped as having no value. (b) It was...
-
Describe the function of each branch of government.
-
A Wheatstone bridge is type of bridge circuit used to make measurements of resistance. The unknown resistance to be measured, Rx, is placed in the circuit with accurately known resistances R1, R2,...
-
The following data from the just completed year are taken from the accounting records of Mason Company: Sales Direct labor cost Raw material purchases Selling expenses Administrative expenses...
-
Read and brief the following cases: a. Willie Nelson Music Co., 85 T. C. 914 b. Independent Contracts, Inc., 73 AFTR2d 94-1406
-
Use activities to calculate the molar solubility of Zn(OH) 2 in (a) 0.0150 M KCl. (b) 0.0250 M K 2 SO 4 . (c) The solution that results when you mix 40.0 mL of 0.250 M KOH with 60.0 mL of 0.0250 M...
-
For a solution in which = 6.5 x 10 -2 , calculate K' sp for (a) AgSCN. (b) PbI 2 . (c) La(IO 3 ) 3 . (d) MgNH 4 PO 4 .
-
What are the five sources of information compiled by media researchers and how are they used in media planning?
-
Repeat the preceding exercise using these letters: RAWHOR. Express all probabilities as fractions
-
When randomly selecting items, if successive selections are made with replacement of previously selected items, which of the five rules of this section apply: (1) fundamental counting rule; (2)...
-
A secretary types 10 different letters and addresses 10 corresponding envelopes. If he is in a hurry and randomly inserts the letters into the envelopes, what is the probability that exactly 9 of the...
-
The winning numbers for the current California Fantasy 5 lottery are 13, 18, 22, 24, and 32 in any order. Do calculations for winning this lottery involve permutations or combinations? Why?
-
Assume that 400 births are randomly selected. Use subjective judgment to determine whether the given outcome is unlikely, and also determine whether it is unusual in the sense that the result is far...
-
Two waves with identical frequency but different amplitudes A 1 = 5.0 cm and A 2 = 3.0 cm, occupy the same region of space (are superimposed). (a) At what phase difference does the resulting wave...
-
Uniform electric field in Figure a uniform electric field is directed out of the page within a circular region of radius R = 3.00 cm. The magnitude of the electric field is given by E = (4.50 x 10-3...
-
Calculate the theoretical cell potential of the following cells. If the cell is short-circuited, indicate the direction of the spontaneous cell reaction. (a) Zn ( Zn2+ (0.1000 M) (( Co2+ (5.87 ( 10-4...
-
Calculate the potential of the following two half-cells that are connected by a salt bridge: (a) A galvanic cell consisting of a lead electrode (right electrode) immersed in 0.0220 M Pb2+ and a zinc...
-
Write balanced net ionic equations to describe (a) The oxidation of Mn2+ to MnO4- by ammonium peroxydisulfate. (b) The oxidation of U4+ to UO22+ by H2O2. (c) The titration of H2O2 with KMnO4.
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
11 The APRN unit director is working at an agency that has a high incidence of medication errors, specifically with heparin. One recent error had a patient receive twice the prescribed dose. Which...

Study smarter with the SolutionInn App


