Calculate the first-order correction to the energy of an electron in electron volts eV, in the ground
Question:
Calculate the first-order correction to the energy of an electron in electron volts eV, in the ground state of hydrogen due to the gravitational potential of the nucleus given by 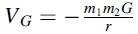 where m1 and m2 are electron and proton masses, respectively, and G is the gravitational constant given by G= 6.672.10-11N.m2.kg-2.
where m1 and m2 are electron and proton masses, respectively, and G is the gravitational constant given by G= 6.672.10-11N.m2.kg-2.
Transcribed Image Text:
VG = - m;m2G
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The firstorder correction to the energy of an electron in the ground state of hydrogen due to the gr...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The kinetic energy of an electron in a particular Bohr orbit of hydrogen is 1.35 10-19 J. (a) Which Bohr orbit does the electron occupy? (b) Suppose the electron moves away from the nucleus to the...
-
The rest energy of an electron is 0.511 MeV. What momentum (in MeV/c) must an electron have in order that its total energy be 3.00 times its rest energy?
-
The de Broglie wavelength of an electron in a hydrogen atom is 1.66 nm. Identify the integer n that corresponds to its orbit.
-
Draw a UML Sequence Diagram for the process involved in paying a vendor for an item in cash. The process to follow is below. [15 points] a) Go to the bank b) Request your account balance c) The bank...
-
Fellowes and Associates Chartered Accountants is a successful mid-tier accounting firm with a large range of clients across Canada. During 2011, Fellowes and Associates gained a new client, Health...
-
Give the basic definitions of oxidation and reduction and explain the basic procedure for balancing redox reactions.
-
If you eat a pound of pasta, will you gain a pound of weight? Why not?
-
Best Practices, Inc., is a management consulting firm. Its Corporate Division advises private firms on the adoption and use of cost management systems. Government Division consults with state and...
-
Suppose the watershed including the San Marcos river can be treated as a 10- kilometer high saturated atmospheric column covering the entire 412 sq. mi. drainage area. With an adiabatic lapse rate of...
-
Use the following narrative to complete the UML class diagram with classes, associations, and multiplicities outlined below and then answer the associated questions: The Nordic Construction Companys...
-
We have seen that in a magnetic field, the magnetic moment of an electron couples to an external magnetic field B to give the so-called Zeeman term H Z =-gB. Bzsz where for free electrons the factor...
-
Equations of motion of an electron in the presence of an electric field. (a) Calculate the velocity of the electron at k = /a. (b) If the electric field E is applied in the -x direction, derive the...
-
At your discount brokerage firm, it costs $9.50 per stock trade. How much money do you need to buy 300 shares of Time Warner, Inc. (TWX), which trades at $22.62?
-
(a) (8 points) Briefly explain, what is a dummy variable? What is the purpose of dummy variables in a regression model? (b) (8 points) How do we interpret regression coefficients associated with...
-
What is nike's output, behavior and input controls for the company and are they affective? Explain
-
Of every 100 units processed, each work station performs as follows: stn#1 = 90 processed correctly the first time and 10 defectives repaired and successfully re-introduced in the process stn#2 = All...
-
1) Tippy Canoe Company sells 500 canoes at $300/each per year and has a contribution margin of $100 per canoe and currently has a net operating income of $49,000 per year. What is their degree of...
-
71. Cox, North, and Lee form a partnership. Cox contributes $189,000, North contributes $157,500, and Lee contributes $283,500. Their partnership agreement calls for a 5% interest allowance on the...
-
Let A be a Hermitian matrix with eigenvalues 1,..., n and orthonormal eigenvectors u1,..., un. Show that A = 1u1u1H + 2u2u2H + ... + nununH
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
How many milliliters of 0.800 M KOH should be added to 5.02 g of 1,5-pentanedioic acid (C 5 H 8 O 4 , FM 132.11) to give a pH of 4.40 when diluted to 250 mL?
-
Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here. H2N NH2 NH2 NH C=0 CH2 CH2 S CH, CH, CH2 CH2 (a) H;NCHCO, (b) H&NCHCO, (c) H,NCHCO, Glutamine Cysteine Arginine
-
(a) Draw the structure of the predominant form (principal species) of 1,3-dihydroxybenzene at pH 9.00 and at pH 11.00. (b) What is the second most prominent species at each pH? (c) Calculate the...
-
Reasons why audiometric testing results may not be representative of noise exposure at workplace?
-
4. Consider the network below. A 5 2 E 8 D B 4 10 You are asked to formulate a mathematical model to determine the path starting from node A, visiting each node only once and returning to node A so...
-
Who would think listening would be the most important conflict management skill?

Study smarter with the SolutionInn App


