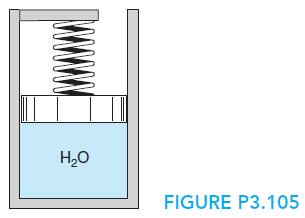
A piston/cylinder arrangement with a linear spring similar to Fig. P3.105 contains R-134a at 60 F, x
Question:
A piston/cylinder arrangement with a linear spring similar to Fig. P3.105 contains R-134a at 60 F, x = 0.6 and a volume of 0.7 ft3. It is heated to 140 F, at which point the specific volume is 0.4413 ft3/lbm. Find the final pressure, the work, and the heat transfer in the process.
Transcribed Image Text:
H20 FIGURE P3.105
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
In order to find the final pressure work and heat transfer in the process we need to use the first ...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
10 kg of water in a piston cylinder arrangement exists as saturated liquid/vapor at 100 kPa, with a quality of 50%. It is now heated so the volume triples. The mass of the piston is such that a...
-
10 kg of water in a piston cylinder arrangement exists as saturated liquid/vapor at 100 kPa, with a quality of 50%. It is now heated so the volume triples. The mass of the piston is such that a...
-
10 kg of water in a piston cylinder arrangement exists as saturated liquid/vapor at 100 kPa, with a quality of 50%. It is now heated so the volume triples. The mass of the piston is such that a...
-
Governmental Funds Statement of Revenues Expenditures and Changes in Fund Balance. You have recently started working as the controller for a small county. The county is preparing its financial...
-
Whether one views homosexual relations as wrong is closely related to whether one views homosexuality as a biological trait or the outcome of one's environment and/or socialization. Thus, it is not...
-
Add NOP instructions to the code below so that it will run correctly on a pipeline that does not handle data hazards. addi $s0, $s1, 5 add $s2, $s0, $s1 addi $s3, $s0, 15 add $s4, $s2, $s1
-
Let \(\tau\) be an initial time which avoids \(\mathbf{F}\)-stopping times. Prove that the \((\mathcal{H})\)-hypothesis holds if and only if \(\alpha_{t}^{u}=\alpha_{t \wedge u}^{u}\).
-
Ron Mitchell is currently working his first day as a ticket seller and cashier at the First Run Movie Theater. When a customer walks up to the ticket booth, Ron collects the required admission charge...
-
Discuss how twenty-first-century marketing is changing the competitive arena. Identify what trends are occurring that are creating change in markets, then: Why do you believe Twitter is changing its...
-
Tayler Smith is a singer who has experienced international success in her career. On May 1, 2017, Tayler started her business, Glitter and Gold Studio where she teaches singing. The following...
-
Water in a piston/cylinder, similar to Fig. P3.225, is at 100C, x = 0.5 with mass 1 kg, and the piston rests on the stops. The equilibrium pressure that will float the piston is 300 kPa. The water is...
-
A 30-ft-high cylinder, cross-sectional area 1 ft 2 , has a massless piston at the bottom with water at 70 F on top of it, as shown in Fig. P3.107. Air at 540 R, volume 10 ft3 under the piston is...
-
Who pays the social security taxes that are levied by the Federal Insurance Contributions Act?
-
Saba gets a year-end bonus in December every year before Christmas. She plans to invest $1000 from the bonus at the end of every year into mutual funds RRSP, which are expected to grow at the rate of...
-
Harold picked up five donuts and four large coffee he paid 6.55. on Tuesday Melinda picked up four donuts and five large coffees she paid 6.77. what id the cost of one donut and one coffee?
-
Stephanie conceives of a compound (QED) in December 2011. She synthesizes the compound in February 2012. Entirely independently of Stephanie, Klay publishes an article in NATURE describing QED on...
-
National Boats manufactures pleasure boats sold to consumers. The boats range in price from $40,000 to $1,500,000. Jake is the president of National Boats. The company provides Jake with one of its...
-
Company G, a calendar year taxpayer, purchased a five-story building on April 2 of the current year for $1 million. The building, which has no historical or architectural significance, was...
-
A refracting telescope has an objective lens with a focal length of 2.20 m and an eyepiece with a focal length of 1.5 cm. If you look through this telescope the wrong way that is, with your eye...
-
Test your confidence in the following Project Decisions: SI. # Question 01 02 03 04 05 06 07 08 09 10 How many years did it take to construct the largest Egyptian Pyramid- Pyramid of Cheops? When was...
-
Identify which of the following compounds is expected to be a stronger base. Justify your choice. N. N.
-
Predict the product of the following reaction, and propose a mechanism for its formation. Na, CH,OH NH;
-
Consider the equilibrium NO 2 (g) NO(g) + 1/2O 2 (g). One mole of NO 2 (g) is placed in a vessel and allowed to come to equilibrium at a total pressure of 1 bar. An analysis of the contents of the...
-
Harry and Megan, two unrelated individuals, are considering forming the Royal Fashion Shoe Corporation in the beginning of 2022. Harry plans to transfer the following assets to the new corporation in...
-
1. What do you identify as the cost driver for the machine change-over costs? Do you agree with the Controller's ultimate classification of these costs as product-sustaining? If not, how would you...
-
The Journal Entry Application's (JEA) function is to create journal entries. The JEA receives notice from the Billing Process Application (BPA) that an invoice has been created. The JEA requests the...

Study smarter with the SolutionInn App


