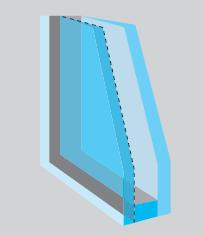
A thermopane window (see Fig. 3.38) traps some gas between the two glass panes. Why is this
Question:
A thermopane window (see Fig. 3.38) traps some gas between the two glass panes. Why is this beneficial?
In Figure 3.38
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The gas has an extremely low conductivity comparativ...View the full answer

Answered By

Gifty Mariya
"I'm a positive individual who has an energetic point of view. I love my work and I get an incredible pride from seeing my understudies create and develop as people. On the off chance that I can decidedly affect their future, I believe I am taking care of my work competently.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A double-glazed window has two panes of glass separated by an air space, Fig. 14-20. (a) Show that the rate of heat flow through such a window by conduction is given by where k1, k2, and k3 are the...
-
Repeat Prob. 1723, assuming the space between the two glass layers is evacuated. Data from 17-23 Consider a 1.2-m-high and 2-m-wide doublepane window consisting of two 3-mm-thick layers of glass ( k...
-
A thermopane window consists of two sheets of glass separated by a volume filled with air (which we will model as N 2 , where = 0.0240 J K -1 m -1 s -1 ). For a thermopane window that is 1 m 2 in...
-
Write a structured flowchart for prob.2.3
-
In this exercise, we will use data from the MTF2011 survey, comparing GPA between two racial/ethnic student groups. First, you'll need to run frequencies of the variable RACE, taking note of what...
-
Suggest a chemical test for distinguishing between (a) [Ni(SO 4 )(en) 2 ]Cl 2 and [NiCl 2 (en) 2 ]SO 4 ; (b) [NiI 2 (en) 2 ]Cl 2 and [NiCl 2 (en) 2 ]I 2 .
-
Prove that if \(X, Y\) are two semi-martingales such that their stochastic logarithms are well defined, then \[\mathcal{L}(X Y)=\mathcal{L}(X)+\mathcal{L}(Y)+[\mathcal{L}(X), \mathcal{L}(Y)]\]
-
Some of Gillettes spokespeople such as Tiger Woods have run into controversy after becoming endorsers for the brand. Does this hurt Gillettes brand equity or marketing message? Explain.
-
A 55 year old employee, earning $50,000 per year is enrolled in only one 401k plan (a traditional 401k). What is the maximum tax deductible contribution she can make to her 401k in 2022?
-
Mary is considering opening a new grocery store in town. She is evaluating three sites: downtown, the mall, and out at the busy traffic circle. Mary calculated the value of successful stores at these...
-
Find the constants in the curve fit for the saturation pressure using Wagners correlation, as shown in the previous problem for water and methane. Find other correlations in the literature, compare...
-
The R-410a in Problem 3.14c is at 1000 kPa, 50C with a mass of 0.1 kg. It is cooled so that the volume is reduced to half the initial volume. The piston mass and gravitation are such that a pressure...
-
Orwell Company accumulates the following adjustment data at December 31. 1. Services provided but not recorded total $1,420. 2. Supplies of $300 have been used. 3. Utility expenses of $225 are...
-
BexTex has a beta of 1.05 and a cost of equity of 13% for the firm overall. The firm is financed solely by common stock. Division A within the firm has an estimated beta of 1.65 and is the riskiest...
-
Suppose that 49th Parallel enters a forward contract to buy 85000 pounds of Arabica coffee at a forward price of $1.7654/pound from a farmer in Ethiopia. What is the contract value for the farmer at...
-
Stock A has a standard deviation of 2.3 and the market portfolio standard deviation is 3.2. The covariance of returns of the market and Stock A is 4.26. What is the correlation coefficient between...
-
What is the required income tax withholding for payment made by government unit to VAT person?
-
The yield of Target Ltd.'s debt is 11%. The leverage ratio and the marcinal tax rate are 3 and 30%, respectively. Comparable Inc., a firm running similar business, has a marginal tax rate of 35%, a...
-
Why does a crystal act as a three-dimensional grating for x-rays but not for visible light?
-
You have just begun your summer internship at Omni Instruments. The company supplies sterilized surgical instruments for physicians. To expand sales, Omni is considering paying a commission to its...
-
The following two compounds each exhibit two heteroatoms (one nitrogen atom and one oxygen atom). In compound A, the lone pair on the nitrogen atom is more likely to function as a base. However, in...
-
2.25 moles of an ideal gas with C V ,m = 5/2 R is transformed from an initial state T 680. K and P = 1.15 bar to a final state T = 298.K and P = 4.75 bar. Calculate U, H, and S for this process.
-
1.10 moles of N 2 at 20.5°C and 6.20 bar undergoes a transformation to the state described by 215°C and 1.75 bar. Calculate ÎS if Jmol-1 -1 + 2.3968 x 10- ? -1.0176 10-3....
-
erences over two periods, Consider an agent (Anna) with pref- U(x1, x2) = u(x1) + Bu(x2) Anna is endowed with 1 unit of x1, and 0 units of x2. Anna faces some interest rate, r, which she takes as...
-
8- Use this data set to answer the following questions. (9 points) 15 11 53 14 36 68 26 15 13 20 a. Find the five-number summary. b. Find the IQR. C. Use IQR to determine whether there are any...
-
The total cost to produce x boxes of cookies is C dollars, where C = 0.0001x3 - 0.04x2 + 2x + 400. In t weeks, production is estimated to be x = 1400 + 100t boxes. (a) Find the marginal cost C'(x)....

Study smarter with the SolutionInn App


