An air compressor has inlet at 100 kPa, 290 K, and brings it to 500 kPa, after
Question:
An air compressor has inlet at 100 kPa, 290 K, and brings it to 500 kPa, after which the air is cooled in an intercooler to 340Kby heat transfer to the ambient 290 K. Assume this first compressor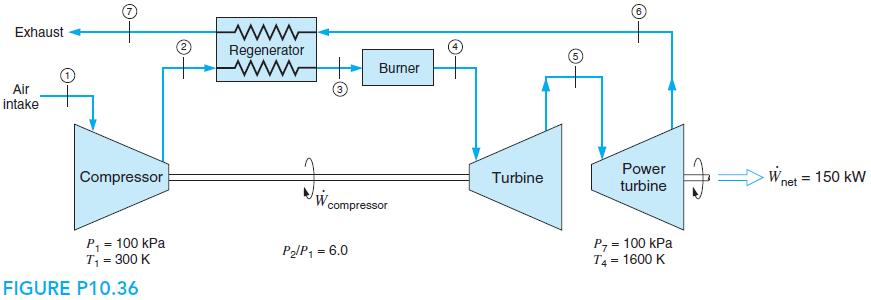
stage has an isentropic efficiency of 85% and is adiabatic. Using constant specific heat, find the compressor exit temperature and the specific entropy generation in the process.
Transcribed Image Text:
Exhaust Regenerator www Burner Air intake Power Compressor Turbine Wnet = 150 kW turbine compressor P, = 100 kPa T, = 300 K P, = 100 kPa T4 = 1600 K PlP, = 6.0 %3D %3D FIGURE P10.36 O- O-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
2 2 The first step is to find the exit temperature of the compressor We can use the first law of the...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
An air compressor has an isentropic efficiency of 0.88. If air at 100 kPa and 25C and a mass ow rate of 20 000 kg/hr is compressed to 500 kPa, what work must be supplied to the compressor? Assume...
-
An air compressor has a significant heat transfer out. See Example 9.4 for how high T becomes if no heat transfer. Is that good or should it be insulated?
-
Air at 600 kPa and 500 K enters an adiabatic nozzle that has an inlet-to-exit area ratio of 2:1 with a velocity of 120 m/s and leaves with a velocity of 380 m/s. Determine (a) The exit temperature...
-
Describe how price variances create incentives to build inventories.
-
Future production requirements in a machine shop call for several automatic bar machines to be acquired to produce three new parts (A, B, and C) that have been added to the shop's product line....
-
What is the molecular formula for each of the following alkenes? (a) C 8 H ? (b) C 10 H ?
-
Why can damage to one side of the cerebrum affect the functioning of the opposite side of the body?
-
Woodhaven Service is a small, independent gas station located in the Woodhaven section of Queens. The station has three gasoline pumps and two service bays. The repair facility specializes in...
-
A stock has a current selling price of $20. The distribution of the price of the stock one year from now is believed to be as follows: PRICE (x) PROBABILITY f(x) 20 .05 21 .06 22 .07 23 .08 24 .09 25...
-
Carla and Scott were married some years ago and have six-year-old twin daughters, Jackie and Kara. In 2015, they divorced. Their divorce decree includes a court order indicating that Carla is to have...
-
A two-stage compressor in a gas turbine brings atmospheric air at 100 kPa, 17C to 500 kPa, then cools it in an intercooler to 27C at constant P. The second stage brings the air to 2500 kPa. Assume...
-
A two-stage air compressor has an intercooler between the two stages, as shown in Fig. P10.35. The inlet state is 100 kPa, 290 K, and the final exit pressure is 1.6 MPa. Assume that the constant...
-
What is the price of a five-year floating-rate note that has coupons at the rate of Libor + 100 bps when the current yield curve is flat at 6%? Assume that the conventions in the market are 30/360...
-
How would Amazon use financial evaluation techniques to decide whether or not to engage in this project: inventory reduction through improved demand forecasting usingpredictive analytics and machine...
-
Steve LOVES coffee. Assume that caffeine levels in human blood serum decay according to the MATH1080 exponential decay model A ( t )= A 0 e - kt . If Steve instantly drinks a coffee, and all of the...
-
If the function f(t) = t(t 21)(t + 1) represents the behavior of a virus during time t and assuming the symptoms started on day 14, how do I use 6 sub-intervals to estimate the total amount of...
-
1) y = sqrt( 1 - x 2 ) a) what is dy/dx ? b) If x increases from 0.6 to 0.6+h where h is some small positive number, approximately how much does y increase by?
-
How do you improve your decision making based on the use of information available? apply example to illustrate a good decision you've made and how information available was part of the process....
-
When a passive activity is sold or otherwise disposed of, what happens to any suspended losses from that activity?
-
What are the key dimensions of critical thinking 2. Watch the NBC Learn video on Diet Scams. What types of claims are made in this video Are they valid Elaborate on your responses. Discuss this video...
-
Compound A has molecular formula C 5 H 10 . Hydroboration- oxidation of compound A produces 2-methylbutan-1-ol. Draw the structure of compound A: Compound A (C,H10) 1) BH, THF 2) H202, NaOH
-
Explain how the ideal gas law can be deduced for the measurements shown in Figures 1.5 and 1.8. Figure 1.5 Figure 1.8 0.1 L 2.5 2.0 1.5 0.2 L 1.0 0.3 L 0.4 L 0.5 L 0.6 L '0.5 -200 100 0 100 200 300...
-
A bowling ball (a) Rolls across a table (b) Falls on the floor. Is the work associated with each part of this process positive, negative, or zero?
-
You want to buy a $26,000 car. You can make a 10% down payment, and will finance the balance with a 5% interest rate for 48 months (4 years). What will your monthly payments be?
-
A disk with mass Mand radius R is on a horizontal table. The disk can rotate about a vertical axis through its centre, O. Figure 1 shows a top view of the disk. A point mass m is attached to the...
-
In the context of an LBO transaction, consider the case of WSP Manufacturing, a company targeted for acquisition. WSP has recently invested in new machinery to increase its production capacity and...

Study smarter with the SolutionInn App


