We want to find the change in u for carbon dioxide between 50C and 200C at a
Question:
We want to find the change in u for carbon dioxide between 50◦C and 200◦C at a pressure of 10 MPa. Find it using ideal gas and Table A.5, and repeat using the B section table.
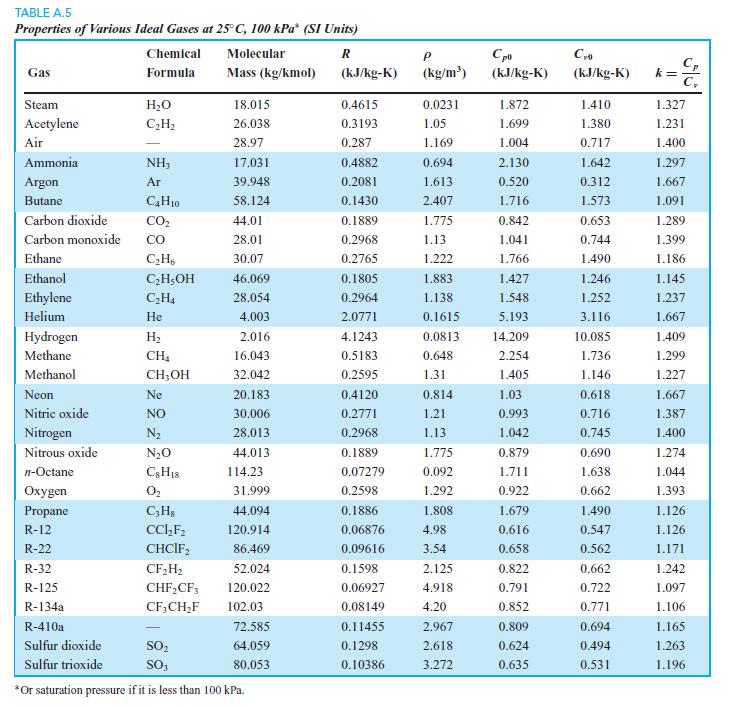
Transcribed Image Text:
TABLE A.5 Properties of Various Ideal Gases at 25° C, 100 kPa* (SI Units) Chemical Molecular C, k = C, Gas Formula Mass (kg/kmol) (kJ/kg-K) (kg/m') (kJ/kg-K) (kJ/kg-K) Steam H2O 18.015 0.4615 0.0231 1.872 1.410 1.327 Acetylene C;H2 26.038 0.3193 1.05 1.699 1.380 1.231 Air 28.97 0.287 1.169 1.004 0.717 1.400 Ammonia NH3 17.031 0.4882 0.694 2.130 1.642 1.297 Argon Ar 39.948 0.2081 1.613 0.520 0.312 1.667 Butane C,H10 58.124 0.1430 2.407 1.716 1.573 1.091 Carbon dioxide CO, 44.01 0.1889 1.775 0.842 0.653 1.289 Carbon monoxide со 28.01 0.2968 1.13 1.041 0.744 1.399 Ethane C;H, 30.07 0.2765 1.222 1.766 1.490 1.186 Ethanol C,H;OH 46.069 0.1805 1.883 1.427 1.246 1.145 Ethylene C,H4 28.054 0.2964 1.138 1.548 1.252 1.237 Helium Не 4.003 2.0771 0.1615 5.193 3.116 1.667 Hydrogen H2 2.016 4.1243 0.0813 14.209 10.085 1.409 Methane CH4 16.043 0.5183 0.648 2.254 1.736 1.299 Methanol CH,OH 32.042 0.2595 1.31 1.405 1.146 1.227 Neon Ne 20.183 0.4120 0.814 1.03 0.618 1.667 Nitric oxide NO 30.006 0.2771 1.21 0.993 0.716 1.387 Nitrogen N2 28.013 0.2968 1.13 1.042 0.745 1.400 Nitrous oxide N20 44.013 0.1889 1.775 0.879 0.690 1.274 n-Octane 114.23 0.07279 0.092 1.711 1.638 1.044 Охудen O2 31.999 0.2598 1.292 0.922 0.662 1.393 Propane C;H3 44.094 0.1886 1.808 1.679 1.490 1.126 R-12 CC,F; 120.914 0.06876 4.98 0.616 0.547 1.126 R-22 CHCIF, 86.469 0.09616 3.54 0.658 0.562 1.171 R-32 CF,H; 52.024 0.1598 2.125 0.822 0.662 1.242 R-125 CHF,CF; 120.022 0.06927 4.918 0.791 0.722 1.097 R-134a CF,CH,F 102.03 0.08149 4.20 0.852 0.771 1.106 R-410a 72.585 0.11455 2.967 0.809 0.694 1.165 Sulfur dioxide SO, 64.059 0.1298 2.618 0.624 0.494 1.263 Sulfur trioxide SO, 80.053 0.10386 3.272 0.635 0.531 1.196 *Or saturation pressure if it is less than 100 kPa.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
u C t From table A5 C 0635 KJ Kg 1 K 1 Aga...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
We want to find the change in u for oxygen gas between 600 K and 1200 K. a) Find it from a constant Cvo from table A.5 b) Find it from a Cvo evaluated from equation in A.6 at the average T. c) Find...
-
We want to find the change in u for oxygen gas between 600 K and 1200 K. a) Find it from a constant Cvo from table A.5 b) Find it from a Cvo evaluated from equation in A.6 at the average T. c) Find...
-
In the following problems we want to find the Fourier transform of the signals. (a) For the signal find its Fourier transform by using the Fourier transform of x(t) = 0.5e at u(t) 0.5e at u(t), a >...
-
A 0.1 cm thick flat copper plate, 2.5 m x 2.5 m square is to be cooled in a vertical position. The initial temperature of the plate is 90?C with the ambient fluid at 30?C. The fluid medium is either...
-
Use the GSS10SSDS file to investigate whether or not Americans have at least two children per person. Use the One Sample T Test procedure to do this test with the variable CHILDS. Do the test at the...
-
The complex [TiCl 6 ] 3 has a ligand field splitting of 160. kJ mol 1 and forms an orange solution. What is the wavelength and color of the absorbed light?
-
The aim of this exercise is to prove that the linear equation \(d Z_{t}=Z_{t^{-}} \mu d M_{t}, Z_{0}=1\) with \(\mu>-1\) has a unique solution. Assume that \(Z^{1}\) and \(Z^{2}\) are two solutions....
-
Multiple-choice questions: a. Which party has the primary responsibility for the financial statements? 1. Bookkeeper 2. Auditor 3. Management 4. Cost accountant 5. None of the above b. Which of the...
-
An annuity pays $500 per year for the next 25 years. If the present value of the annuity is $7,500 today what is the annual rate of return promised by the annuity?
-
A firm is considering using the Internet to supplement its traditional sales methods. Using data from an industry association, the firm estimates that 1 of every 1,000 Internet hits results in a...
-
Estimate the constant specific heats for R-134a from Table B.5.2 at 100 kPa and 125C. Compare this to the specific heats in Table A.5 and explain the difference.
-
For a special application, we need to evaluate the change in enthalpy for carbon dioxide from 30C to 1500C at 100 kPa. Do this using the constant specific heat value from Table A.5 and repeat using...
-
Percentage-of-completion and completed contract methods of income recognition. Raytheon has agreed to construct a high-tech missile detection system for $900 million. Expected and actual costs to...
-
Below you are given two machines that if adopted would lower your annual production cost. Your "required rate of return (discount rate)" is 11.5%. Machine A After-Tax Initial Cost = $3200 3-year Life...
-
Gerald owns a specialty printing company. He is losing sales to a competitor because he does not offer any credit and has decided that he will open credit lines to his customers. What is the...
-
A sold a parcel of land to B for Php20,000.00. In the deed of sale, there is a stipulation that the purchase price shall be paid on a certain date and that in case of failure to pay on such date, A...
-
Stuart is thinking about retiring soon, as he has been saving his entire working life.To maintain his lifestyle, he needs to get $80,000 per year in income from his investments.If he wants to plan to...
-
Vince has a coin collection that he estimates is worth $950 today.Based on what he has seen in the past, he is confident that the value of this coin collection would increase to $2,000 in 5...
-
Explain, using Huygens's principle, why the Poisson spot is expected.
-
SCHEDULE OF COST OF GOODS MANUFACTURED The following information is supplied for Sanchez Welding and Manufacturing Company. Prepare a schedule of cost of goods manufactured for the year ended...
-
At the transition temperature of 95.4C, the enthalpy of transition from rhombic to monoclinic sulfur is 0.38 kJ mol 1 . a. Calculate the entropy of transition under these conditions. b. At its...
-
One mole of a van der Waals gas at 25.0????C is expanded isothermally and reversibly from an initial volume of 0.010 m 3 to a final volume of 0.095 m 3 . For the van der Waals gas, (U/V)T = a/V 2 m ....
-
From the following data, derive the absolute entropy of crystalline glycine at T = 300.K. You can perform the integration numerically using either a spreadsheet program or a curve-fitting routine and...
-
The bell tower of the cathedral in Pisa, Italy, leans 5.6 from the vertical. A tourist stands 104 m from its base, with the tower leaning directly toward her. She measures the angle of elevation to...
-
How many hours would it take for 500 mL of intravenous (IV) solution to be delivered if it is flowing at 60 drops/ minute? This set is calibrated at 100 drops per mL.
-
The limit of the Riemann sum n 183-3 n lim 818 n i=1 can be expressed as a definite integral in many different ways. Find two of them with the limits of integration specified below. (a) (b) S 0 3 1...

Study smarter with the SolutionInn App


