An open settling tank shown in the figure contains a liquid suspension. Determine the resultant force acting
Question:
An open settling tank shown in the figure contains a liquid suspension. Determine the resultant force acting on the gate and its line of action if the liquid density is 850 kg/m3.
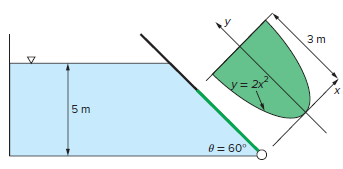
Transcribed Image Text:
3 m y= 2x 5 m 0 = 60°
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
An open settling tank contains liquid suspension The resultant force acting on the gate and ...View the full answer

Answered By

Muhammad Rehan
Enjoy testing and can find bugs easily and help improve the product quality.
4.70+
10+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Thermal-Fluid Sciences
ISBN: 978-0078027680
5th edition
Authors: Yunus A. Cengel, Robert H. Turner, John M. Cimbala
Question Posted:
Students also viewed these Physics questions
-
Determine the resultant force acting on the 0.7-m-high and 0.7-m-wide triangular gate shown in Fig. P11??15 and its line of action. 0.3 m. 0.7 m 0.9 m Water 0.7 m
-
From Prob. 11-22, knowing that the density of the suspension depends on liquid depth and changes linearly from 800 kg/m3 to 900 kg/m3 in the vertical direction, determine the resultant force acting...
-
If the resultant force acting on the bracket is to be FR = {800j} N, determine the magnitude and coordinate direction angles ofF. 45 = 750 N
-
As our energy structure transitions toward renewable fuels, forest-based biomass fuels benefit from this transition. What are the likely effects of this transition on consumers, producers, and the...
-
A water tank springs a leak. Find the speed of water emerging from the hole if the leak is 2.9 m below the surface of the water, which is open to the atmosphere?
-
A compound X (C10H14O) dissolves in aqueous sodium hydroxide but is insoluble in aqueous sodium bicarbonate. Compound X reacts with bromine in water to yield a dibromo derivative, C10H12Br2O. The...
-
Consider the data below. These data were collected in an experiment where \(x_{1}=\) reaction time in minutes and \(x_{2}=\) temperature in degrees Celsius. The response variable \(y\) is...
-
Peters Restaurants acquired Corner Diners. The financial records of Corner Diners included the following: Book Value of Assets...
-
If you have $577 today and you think you can earn an interest rate of 6% per year, how many years until you have $982?
-
Macon Controls produces three different types of control units used to protect industrial equipment from overheating. Each of these units must be processed by a machine that Macon considers to be...
-
A cylindrical tank is fully filled with water (Fig. P11??21). In order to increase the flow from the tank, an additional pressure is applied to the water surface by a compressor. For P 0 = 0, P 0 = 3...
-
The following 2.5 m 8.1 m 6 m tank shown is filled by oil of SG = 0.88. Determine (a) The magnitude and the location of the line of action of the resultant force acting on surface AB (b) The...
-
Mitch Ziegel and Rosie Punter are discussing the audit plan for a large manufacturing company. The company has two main manufacturing plants plus several warehouse and distribution centres (one in...
-
1. Determine the capitalized cost of an equipment costing P2M with and annual maintenance of P200,000.00 if money is worth 20% per annum. 2. A dam will have a first cost of $5,000,000 an annual...
-
solve this question. Option A: Life = 6 years, Initial cost = $28,000, Annual operating cost = $2,000 Option B: Life = 3 years, Initial cost = $23,000, Annual operating cost = $3000 MARR = 9% n=3...
-
What is the most reasonable estimate of pH for a 5.5x108 M solution of NaOH? a. 7.3 b. 6.7 IXID" = [H] [[SH] c. 7.1 d. 6.9 e. 7.0 3,5 kiCR= [Nu] X + [OH-] (N+][#] = [C#] 5.5810) [HT] = [0#]
-
A chemical substance which facilitates the transport of nerve impulses among nerve cells is known as a(n) O aspirin O analgesic histamines O neurotransmitters antipyretic
-
The voltages versus Li measured in Li-Co-O system are shown in the ternary phase diagram. The Gibbs free energies of pure substances (unit kJ/mol are G(Li) -8.67, G(LiO)=-610.027, G(COO)=-253.737,...
-
State University is scheduling 24 sections of a large computer skills course in the Fall semester. There are eight time slots for these sections, four on Monday/ Wednesday (MW) and four on...
-
The following table shows the rates of total return in successive years from 2004 to 2008 for the Sprott Canadian Equity Fund and for the benchmark Toronto Stock Exchange S&P/TSX Composite Index. By...
-
Can a tank with the outflow rate fixed by a constant speed pump reach a steady state if the inlet flow rate undergoes a step change? Why, or why not? If the transfer function is G(s) = K/s, is it...
-
An additive process model is depicted in the figure below. For (unit impulse) (a) Derive the response Y(s) and describe y(t) quantitatively. (b) Simulate the response and identify its major...
-
For a stirred-tank heater, assume the transfer function between the heater input change u(t) (cal/sec) and the tank temperature change y(t)( C) can be modeled as G(s) = 5 / 3s + 1 (a) Using the...
-
If you weigh your portfolio in 80% risky and 20% risk-free t bills. The optimal risky portfolio consists of 40% in bonds and 60% in ETFs. How much will you have invested in t-bills, bonds, and etfs?
-
b. A 25-year mortgage loan of $184,000 at a 10 percent compound annual interest rate with equal installment payments at the end of each year.
-
What are Cloud-based Services? How does this differ to the common virtualization model?

Study smarter with the SolutionInn App


