Indicate the molecular geometry around each carbon atom in the compound H;CCH=CHCH,CCH,COOH
Question:
Indicate the molecular geometry around each carbon atom in the compound
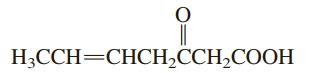
Transcribed Image Text:
H;CCH=CHCH,CCH,COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Numbering the seven carbon atoms from left to right the ...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following samples has a larger mass? O 19.3 g of Au 8.92 10 atoms of Pt Identify the molecular geometry around the three selected atoms. Treat that atoms as the central atom for each...
-
Search Textbo a. chloroform, CHCI3 (carbon is central atom) Lewis structure Total number of electron groups around the central atom Electron geometry Molecular shape b. Carbon tetrachloride, CCI4...
-
The lactic acid molecule, CH3CH (OH) COOH, gives sour milk its unpleasant, sour taste. (a) Draw the Lewis structure for the molecule, assuming that carbon always forms four bonds in its stable...
-
What is the coefficient of sliding friction and what is a representative value for this coefficient for the brittle crust?
-
A coagulation-microfiltration process for removing bacteria from water was investigated in Environmental Science & Engineering (Sept. 1, 2000). Chemical engineers at Seoul National University...
-
Clayton participates in his employers nonqualified deferred compensation plan. For 2022, he is deferring 10 percent of his $320,000 annual salary. Assuming this is his only source of income and his...
-
What is the difference between domain definition and domain dictionary? Give an example for each.
-
You are provided with the following information for Perkins Inc. for the month ended October 31, 2014. Perkins uses a periodic method for inventory. Instructions (a) Calculate (i) ending inventory ,...
-
Chang Company provided the following data for this year: Manufacturing overhead costs incurred: Indirect materials Indirect labor Property taxes, factory Utilities, factory Depreciation, factory...
-
1. Each catalog costs approximately $2 to mail (including printing, postage, and mailing costs). Estimate the gross profit that the firm could expect from the remaining 180,000 names if it selects...
-
Draw the cis and trans isomers of the compound 3-methyl-3-hexene.
-
Complete and balance the reaction for each of the following undergoing a combustion reaction. a. 2-methylpentane b. 2,2-dimethylpropane c. 3-methyl-3-hexene d. 3-ethyl-3-propanol e. ethylbenzene
-
List the FMS layers and describe their functions.
-
As a result of a community-wide salary survey, five months into the new fiscal year, all coders were given a 3.5% raise to bring their wages into parity with the surrounding area hospitals. This wage...
-
Jenkins Manufacturing produces a wide variety of plastic products, mostly for major department stores. They are gearing up for production of their deluxe lawn sprinkler. Currently, they can produce...
-
How has the Financial Reporting Council (FRC) in the UK contributed to shaping the history and current regulatory environment of external auditing in the UK and in the world? Discuss key initiatives...
-
Identify a difference between incentives and merit adjustments. what is the difference in both points. Explain
-
Very Light Jets (VLJs) are one-pilot, two-engine jets that weigh 10,000 pounds or less and have only five or six passenger seats. Since they cost half as much as the most inexpensive business jets,...
-
Wild Bird Feeders produces deluxe bird feeders for distribution to catalogue companies and wild bird stores. The company uses an absorption costing system for internal reporting purposes but is...
-
Charles owns an office building and land that are used in his trade or business. The office building and land were acquired in 1978 for $800,000 and $100,000, respectively. During the current year,...
-
a. When 0.10 mol of the ionic solid NaX, where X is an unknown anion, is dissolved in enough water to make 1.0 L of Concept Explorations solution, the pH of the solution is 9.12. When 0.10 mol of the...
-
Which of the following beakers best represents a container of a weak acid, HA, in water? (Water molecules have been omitted for clarity.) ,- = A- = HA A
-
You have 0.10-mol samples of three acids identified simply as HX, HY, and HZ. For each acid, you make up 0.10 M solutions by adding sufficient water to each of the acid samples. When you measure the...
-
The community nursing service team provides nursing care to patients in the community, Discuss?
-
Why are angiosperms so diverse (when compared with non-flowering lineages of plants)? How expensive flowers are to produce for the plant (they cost lots of ATP to make); what benefit do they provide...
-
Martinez Company's relevant range of production is 7,500 units to 12,500 units. When it produces and sells 10,000 units, its average costs per unit are as follows: Average Cost Per Unit Direct labor...

Study smarter with the SolutionInn App


