Given below are four proposed modifications of the van der Waals equation of state. Are any of
Question:
Given below are four proposed modifications of the van der Waals equation of state. Are any of these modifications reasonable? Explain carefully; statements such as, “It isn’t cubic in volume” do not qualify.
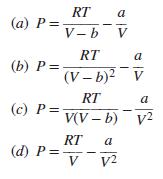
Transcribed Image Text:
RT (а) Р%3 a V- b V RT a (b) P = (V – b)? V RT a (c) P= V(V – b) v2 RT (d) P = V a V2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
The van der Waals equation of state is For carbon dioxide, a = 0.3640 Pa m 6 mol 1 and b = 4.267 Ã 10 5 m 3 mol 1 . Find the pressure of 0.7500 mol of carbon dioxide if V = 0.0242 m 3 and T =...
-
The van der Waals equation of state (Equation 5.3-6) is to be used to estimate the specific molar volume V (L/mol) of air at specified values of T (K) and P(atm). The van der Waals constants for air...
-
The van der Waals equation of state (Equation 5.3-7) is to be used to estimate the specific molar volume V(L/mol) of air at specified values of T(K) and P(atm). The van der Waals constants for air...
-
The National Health Statistics Reports dated Oct. 22, 2008, included the following information on the heights (in.) for non-Hispanic white females: a. Calculate and interpret a confidence interval at...
-
Is a firm justified in paying a bribe if it believes a competitor will do so to win an important contract? Explain.
-
Pioneer Parts, a manufacturer of windows for commercial buildings, reports the following account information for last year (all costs are in thousands of dollars): Information on January 1...
-
When do we use sub-flows?
-
Ray County administers a tax custodial fund, an investment trust fund, and a private-purpose trust fund. The tax custodial fund acts as custodian for the county, a city within the county, and the...
-
How does Bayesian inference play a role in the quantification of model risk within advanced risk management frameworks ?
-
Sarah is the sole owner of Bluegrass Corporation. The basis and value of her stock investment in Bluegrass are approximately $100,000. In addition, she manages Bluegrass's operations on a full-time...
-
With reference to Prob. 2.47, assume air to be an ideal gas, and develop an expression giving the household air temperature as a function of time. Problem 2.47 The heating of a home to increase its...
-
For a steady-flow heat exchanger with a feed temperature of 100C, compute the outlet stream temperature when heat in the amount of 12 kJmol 1 is added to the following substances. (a) Methane (b)...
-
The Railway Labor Act and the National Labor Relations Act govern most all aspects of air transport collective bargaining. Identify the differences between these two acts as to: a) Contracts b)...
-
In what ways do developing countries depend on rich countries? In what ways is the opposite true?
-
A Job Creation through Entrepreneurship Act, debated in the House of Representatives in 2009, would award grants to small business owners, some of which would be aimed at women, Native Americans, and...
-
Explain the concept of urban bias. What policies are associated with it, and what are their likely effects on urban and rural areas?
-
Which of the following are components of the opportunity cost of being a full-time student? The cost of Tuition and books Residence and a meal plan A subscription to the New Yorker magazine The...
-
What were the central findings of Melissa Dells research on the mita system, and what is their significance for the study of economic development?
-
(a) Determine whether is in the span of (b) Is In the span of and (c) Is in the span of and 123 011 110 121 120 3 3012 0130 1201 2011
-
Representative data read from a plot that appeared in the paper Effect of Cattle Treading on Erosion from Hill Pasture: Modeling Concepts and Analysis of Rainfall Simulator Data (Australian Journal...
-
Calculate the theoretical flame temperature when ethylene at 25(C is burned with: (a) The theoretical amount of air at 25(C. (b) 25% excess air at 25(C. (c) 50% excess air at 25(C. (d) 100% excess...
-
For steady Bow through a heat exchanger at approximately atmospheric pressure, what is the final temperature, (a) When heat in the amount of 800 kJ is added to 10 mol of ethylene initially at 200(C?...
-
For steady Bow through a heat exchanger at approximately atmospheric pressure, what is the final temperature, (a) When heat in the amount of 800 kJ is added to 10 mol of ethylene initially at 200(C?...
-
2. A project requires $150 million investment. If you invest today, the present value of future cash flow is worth $180 million. You can wait for one year to observe the demand of the market;...
-
Respond to this students posting. In your response, you may want to comment on the ethical issues that arise in different career fields or give suggestions for dealing with the ethical questions that...
-
What does the Hedonic View of happiness primarily focus on?

Study smarter with the SolutionInn App


