Convert the following perspective formulas to Fischer projections. (a) (b) (c) (d) H OH .C. CH; CH,OH
Question:
Convert the following perspective formulas to Fischer projections.
(a)
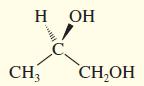
(b)
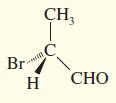
(c)
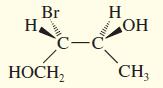
(d)
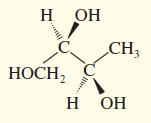
Transcribed Image Text:
H OH .C. CH; CH,OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
1 2 3...View the full answer

Answered By

Vartika ..
Having secured 10 CGPA during school level education, and being one of the toppers in my school as well as being college topper with distinction, I have always conceptualised chemistry not just as a subject, but also as a mystery to explore more about.
I have been teaching since college days and have helped students clear their doubts as well as exams.
The biggest compliment i received was when one of my students said that I answer even those queries, which even their teachers couldn't answer.
I welcome you all to come with me and explore the mysterious world of chemistry ;)
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw enantiomers for each of the following compounds using: a. perspective formulas b. Fischer projections CH CH 1. CHjCHCH2H 2. CICH2CH2CHCH2CH3 3. CHjCHCHCHj
-
Convert the following Fischer projections to perspective formulas. (a) (b) (c) (d) H,N- H- CH;
-
Convert the following Haworth projections to a Fischer projection. Name the monosaccharides you have drawn. a) CH OH
-
In Exercises use a graphing utility to graph the curve represented by the parametric equations. Indicate the direction of the curve. Identify any points at which the curve is not smooth. Curtate...
-
Why does buffer capacity increase as the concentration of buffer increases?
-
What would be the effect of each of the following on Uptown Banks excess reserves and loan-making ability if the bank had $600 million in deposits, a 5 percent reserve requirement, and actual...
-
Sarah serves as Chief Executive Officer (CEO) for Biostate Corporation. Biostate is a pharmaceutical company that conducts research on the development of new cancer drugs. It has developed several...
-
Ken owns and operates a famous candy store and makes most of the candy sold in the store. Business is particularly heavy during the Christmas season. Ken contracts with Sweet, Inc., to purchase ten...
-
1. How the singly linked lists can be represented? 2. How the doubly linked list can be represented? 3. What are benefits of ADT? 4. When singly linked list can be represented as circular linked...
-
Zeff Company purchases a delivery van on January 1, Year 1, at a cost of $15,849. It has a useful life of four years and no estimated salvage value . When making the purchase decision, the company...
-
A chiral sample gives a rotation that is close to 180. How can one tell whether this rotation is +180 or -180?
-
Give the stereochemical relationships between each pair of structures. Examples are same compound, structural isomers, enantiomers, diastereomers. Which pairs could you (theoretically) separate by...
-
Beam AD carries the two 40-lb loads shown. The beam is held by a fixed support at D and by the cable BE which is attached to the counter-weight W. Determine the reaction at D when (a) W = 100 lb, (b)...
-
The Assembly Department of the Long - Distance Golf Club Super Driver Company, has the following production and cost data at the end of March, 2 0 2 1 . Production: 1 2 , 6 0 0 units started into...
-
The 1,500 m3 concrete loading ramp is redesigned from a concrete strength of 35MPa to the lower strength of 25MPa (cost changed from $215 per m3 to $190 per m3). What is the net adjustment for the...
-
3. Here is an image of the communication between the three processes according to their local clocks: pl p2- p3- m local time local time local time Which x y communications require time compensation...
-
If the manufacturer began the month with $ 3 , 1 0 0 in raw materials inventory, compute the ending balance in the Raw Materi Inventory account at the end of the period.
-
You have received a job offer with total compensation of $70,000 per year from a mature company. You have also received a job offer to earn $90,000 per year from a start-up. You are willing to take...
-
The drawing shows an equilateral triangle, each side of which has a length of 2.00 cm. Point charges are fixed to each corner, as shown. The 4.00 C charge experiences a net force due to the charges...
-
In Exercises 105108, evaluate each expression without using a calculator. log(ln e)
-
Predict the relative intensities of the three peaks in the mass spectrum of dichloromethane at m/z = 84, 86, and 88.
-
From the molecular masses and the relative intensities of their M and M + 1 peaks, suggest molecular formulas for the following compounds. M (m/z = 82; 37%), M + 1 (2.5%); contains C and H.
-
Suggest a structure for each of the ions corresponding to the following peaks in the EI mass spectrum of ethyl bromide, and give a mechanism for the formation of each ion. (The numbers in parentheses...
-
Exactly 24 months ago in 2019, centronics inc issued a 15 year bond. the bond was issued at par to yield 6%. it pays interest annually. over the last 12 months, yields on bonds of similar risk have...
-
How COVID-19 news evoked sentiments and emotions. What are the financial implications of these sentiments for investors/policy makers?
-
How does the network of global stock markets reacts to covid-19? What countries (stock markets) plays most important role during covid-19? Explain.

Study smarter with the SolutionInn App


