Draw a graph, similar to Figure 3-9, of the torsional strain of 2-methylpropane as it rotates about
Question:
Draw a graph, similar to Figure 3-9, of the torsional strain of 2-methylpropane as it rotates about the bond between C1 and C2. Show the dihedral angle and draw a Newman projection for each staggered and eclipsed conformation.
Figure 3-9
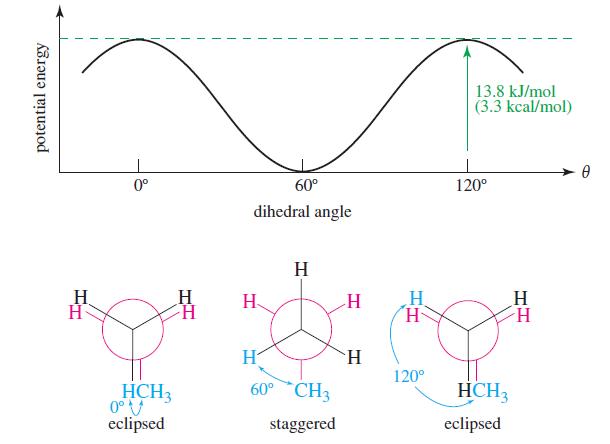
Transcribed Image Text:
13.8 kJ/mol (3.3 kcal/mol) 0° 60° 120° dihedral angle H H. H H H. H H. 120° 60° CH3 HCH3 HCH3 0°V eclipsed staggered eclipsed potential energy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (20 reviews)
From the figure we get the value of one CH 3 H interaction value whi...View the full answer

Answered By

Supriya Mahata
Q and A expert in Chegg , Course hero and BARTLBY
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a graph, similar to Figure 3-11, of the torsional energy of 2-methylbutane as it rotates about the C2¬C3 bond. Figure 3-11 3.8 kJ (0.9 kcal) 15 kJ 21 kJ (3.6 kcal) (5 kcal) -21 kJ (5 kcal)...
-
Construct a graph, similar to Figure 3-11, of the torsional energy of 3-methylpentane along the C2-C3 bond. Place C2 in front, represented by three bonds coming together in a Y shape, and C3 in back,...
-
Prednisolone acetate is an anti-inflammatory agent in clinical use. It is similar in structure to cortisol, with the following two differences: (1) Prednisolone acetate exhibits a double bond between...
-
In Exercises find a power series for the function, centered at c and determine the interval of convergence. f(x) 2 6 - x' C = -2
-
(a) Write the chemical reactions whose equilibrium constants are Kb and Ka for imidazole and imidazole hydrochloride, respectively.
-
Consider the single replicate of the 2 4 design in Example 6-2. Suppose that we ran five points at the center (0, 0, 0, 0) and observed the following responses: 73, 75, 71, 69, and 76. Test for...
-
A dicing system has a first cost of $\$ 68,000$ with a life of 8 years and a salvage value of probably not more than $\$ 9,000$. The corporate MARR is $14 \%$. If the system has a maintenance cost of...
-
On January 1, 2008, Ameen Company purchased a building for $36 million. Ameen uses straight-line depreciation for financial statement reporting and MACRS for income tax reporting. At December 31,...
-
Havana Hats makes the world's best hats. Information for the last eight months follows: Number of Hats Month January Produced Total Cost 1,800 $ 9,000 February 4,890 15,648 March 2,300 10,350 April...
-
The liabilities of Organic Foods are made up of $60,000 in notes payable as of its December 31 year-end. For those notes payable, $3,000 is due within the next year. Prepare the liabilities section...
-
Give the structures of 4-isopropyloctane and 5-tert-butyldecane.
-
Write structures for a homologous series of alcohols (R-OH) having from one to six carbons.
-
Complete the following table by calculating the missing entries and indicating whether the solution is acidic or basic.
-
Why is paying attention to an interviewers verbal and nonverbal cues as important as responding to questions?
-
Discuss why it could be useful to look for stepping-stone opportunities alongside specific job positions. Explain your answer.
-
How can design consistency be useful in the setting of presentations of company products and branding to customers?
-
Assume you are a news anchor for a television channel presenting election results from different states or districts as well as tweets from your viewers. Would a linear or a nonlinear presentation be...
-
Students may well be asked to discuss their leisure pursuits during selection processes. Your task: Prepare a brief presentation about one of your leisure pursuits. Your purpose should be to...
-
On January 1, Poitras Ltée, a public company, purchases 20% of Hook Corporation's common shares for $250,000 for strategic purposes. For the year ended December 31, Hook reports profit of...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
Give the structures of all epoxides that could in principle be formed when each of the following alkenes reacts with meta-chloroperoxybenzorc acid (mCPBA). Which epoxide should predominate in each...
-
Give the structures of all epoxides that could in principle be formed when each of the following alkenes reacts with meta-chloroperoxybenzorc acid (mCPBA). Which epoxide should predominate in each...
-
When the naturally occurring amino acid (S)-methionine is converted into methionine sulfoxide, two isomers with different physical properties are formed. What are their structures and what is their...
-
Use the following to answer questions 34-36 The year-end adjusted trial balance of the Corporation included the following account balances: Retained earnings $200,000 Service revenue 600,000 Salaries...
-
Jackie Co., a 90% owned subsidiary of Nick Inc., sold land to Nick on May 1, 2024, for $80,000. The land originally cost Jackie $85,000. Jackie reported net income of $200,000, $180,000, and $220,000...
-
The most recent financial statements for AppleBanana Co. are shown here: Income Statement Sales Costs Balance Sheet $ 200,000 130,000 Current assets Fixed assets $ 120,000 280,000 Debt Equity $...

Study smarter with the SolutionInn App


