Show how you might synthesize the following tertiary amine three different ways, each using a different secondary
Question:
Show how you might synthesize the following tertiary amine three different ways, each using a different secondary amine and adding the final substituent by
(a) Reductive amination (3 ways).
(b) Acylation–reduction (3 ways).
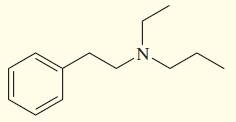
Transcribed Image Text:
N.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
aCondensation of the secondary amine with a carbonyl group ie an ald...View the full answer

Answered By

Thiruvengadam Vedamurthy
I have completed MPhil in Organic chemistry and MSc in chemistry. I am teaching for X, XI and XIIth CBSE as well as state board syllabus for more than two years. Currently , I am teaching chemistry for the NEET competitive examination.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how you might synthesize each of the following compounds using, as your starting materials, esters, ketones, acyl halides, and so on: (a) (b) (c) OEt OEt
-
Show how you might synthesize each of the following compounds from 1-butanol: (a) Butylamine (free of 28 and 38 amines) (b) Pentylamine (c) Propylamine (d) Butylmethylamine
-
Show how you might synthesize each of the following starting with a-tetralone (Section 15.9): (a) (b) (c) (d) HO So OH 02 H5
-
Bryce owns 200 shares of Basic Company stock that he purchased for $8,000 three years ago. On December 28, 2021, Bryce sold 100 shares of the stock for $2,500. On January 3, 2022, Bryce repurchased...
-
Explain why the total solubility of lead in Figure 6-3 first decreases and then increases as [ I-] increases. Give an example of the chemistry in each of the two domains.
-
On July 1, 20X1, McVay Corporation issued $15 million of 10-year bonds with an 8% stated interest rate. The bonds pay interest semiannually on June 30 and December 31 of each year. The market rate of...
-
How would a company know that its product or service was preferred in the marketplace? What could a competitor do to erode this favorable position?
-
Perry Co. predicts it will use 25,000 units of material during the year. The expected daily usage is 200 units, and there is an expected lead time of five days and a desired safety stock of 500...
-
es Determine the ending balance of each of the following T-accounts. Accounts Payable Cash 140 90 2,400 340 100 3,300 60 540 10 Supplies 10,400 1,500 4,200 9,200 Accounts Receivable 800 190 190 190...
-
HERE COME THE CLOWNS! Is the name of a traveling circus? The ledger accounts of the business at June 30, 2011, are listed here in alphabetical order: Instructions a. Prepare a balance sheet by using...
-
A compound of formula C 11 H 16 N 2 gives the IR 1 H NMR, and 13 C NMR spectra shown. The proton NMR peak at 2.0 disappears on shaking with D 2 O Propose a structure for this compound, and show how...
-
Propose mechanisms for the nucleophilic acyl substitutions to form ethyl benzoate and N-methylacetamide as shown on the previous page.
-
How does phosphorus that forms, relatively insoluble inorganic compounds in soils find its way into streams and other waterways?
-
Try creating a text cloud from a lecture that one of your teachers gives. Take notes on the lecture or use an audio recorder and then transcribe the recording. Structure your notes or the...
-
If the length of the major axis of the earth's orbit is \(186,000,000 \mathrm{mi}\) and its eccentricity is 0.017 , how far is the earth from the sun when it is at aphelion and at perihelion?
-
Using Schein's definition of organizational culture from the section that explains approaches to analyzing workplace cultures, divide the class into six teams and assign one part of the definition to...
-
Concerning the decision-making processes of people in workplaces, Mary Douglas (1986, 4) asserts that "an answer is only seen to be the right one if it sustains the institutional thinking that is...
-
If you are a practicing technical communicator currently, find some document in your organization that you can talk about not only for what it says, but also for what it does. What kind of cultural...
-
Assume that Stoney Inc. sold bonds with a face value of $100,000 for $102,000. Was the market interest rate equal to, less than, or greater than the bonds' contractual interest rate? Explain.
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
Give the principal product(s) expected when 4-methyl-cyclohexene or other compound indicated reacts under the conditions in Problem 17.18. (a) Br2 in CH2Cl2, dark (b) A-bromosuccinimide in CCl4,...
-
Identify the benzylic carbons in the following structures. H,C-
-
A student Al Lillich has prepared a pure sample of 3-bromo-l-butene (A). Several weeks later he finds that the sample is contaminated with an isomer B formed by allylic rearrangement. (a) Give the...
-
The trial balance for a company listed the following account balances at December 31, Year 1, the end of its fiscal year: cash, $36,000; accounts receivable. $31,000; Inventory, $45,000; equipment...
-
A corporation issues 13 %, 15-year bonds with a par value of $570,000 and semiannual interest payments. On the issue date, the annual market rate for these bonds is 11%, which implies a selling price...
-
A production department reports the following conversion costs. Equivalent units of production for conversion total 436,000 units this period. Calculate the cost per equivalent unit of production for...

Study smarter with the SolutionInn App


