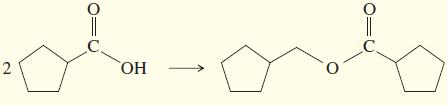
Show how you would accomplish the following multistep syntheses. You may use any additional reagents and solvents
Question:
Show how you would accomplish the following multistep syntheses. You may use any additional reagents and solvents you need.
a. PhCH2CH2OH → PhCH2CH2COOH
b.
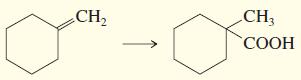
c.
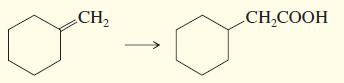
d.
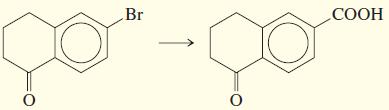
e.
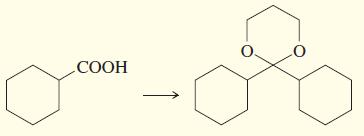
f.
Transcribed Image Text:
CH3 CH2 СООН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a b c d e f Ph...View the full answer

Answered By

AKASH PANJA
I'm currently in 4th semester of Msc organic chemistry. I'm doing MSc from Central University Of Haryana, India. I've done two research Projects from Centre Of Biomedical Research (CBMR), Lucknow, India in organocatalysis and metal Catalyzed Asymmetric synthesis.
I'm a former subject expert in chegg. I can solve any questions of chemistry with a very detailed explanations in a cheap rate compare to others.
https://www.linkedin.com/in/akash-panja-516247196
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how you would accomplish the following synthetic conversions. You may use any additional reagents and solvents you need. (a) (b) (c) (d) Ph-CHO OH PhCHOPhCHC CCH2CH3
-
Show how you would accomplish the following multistep conversions. You may use any additional reagents you need. a. b. c. d. dimethyl adipate and allyl bromide
-
Show how you would accomplish the following syntheses. You may use whatever additional reagents you need. (a) (b) (c) (d) (e) (f) CHO CH,OH CHO CHO CH,Br Ph of CH, CH cth CH CH CHC-Ph 0 BrCH,CH,CCH...
-
Use any method to determine if the series converges or diverges. Give reasons for your answer. n10 10" n=1
-
Find [Cu2+] in a solution saturated with Cu4(OH)6(SO4) if [OH-1] is fixed at 1.0 10-6M. Note that Cu4(OH)6(SO4) gives 1 mo of SO42- for 4 mol of Cu2+. Ksp = 2.3 10-69
-
Information concerning the capital structure of the Petrock Corporation is as follows: During 20X1, Petrock paid dividends of $1 per share on its common stock and $2.40 per share on its preferred...
-
What is the purpose of financial statements? Would you want to produce them even if they were not required, say, for entity tax reporting?
-
In 2013, Lisa Perry opened Lisas Jeans Company, a small store that sold designer jeans in a suburban mall. Perry worked 14 hours a day and controlled all aspects of the operation. The company was...
-
IBM Corporation was the leading manufacturer of electronic computers in the world. Based on its internal memoranda, IBM's long-run total cost of producing various quantities of its Pisces (370/168)...
-
E3.6 Find Vo in Fig. E3.6 using nodal analysis.
-
When pure (S)-lactic acid is esterified by racemic butan-2-ol, the product is 2-butyl lactate, with the following structure: (a) Draw three-dimensional structures of the two stereoisomers formed,...
-
The following NMR spectra correspond to compounds of formulas (A) C 9 H 10 O 2 , (B) C 4 H 6 O 2 , and (C) respectively. Propose structures, and show how they are consistent with the observed...
-
Some hospitals are beginning to use automated pharmacies, a large machine that can dispense common medications like an ATM. What are the benefits and challenges of this technology?
-
Your test scores are \(78,95,87\), and 92. What score on the fifth exam will make your average score at least 80 ?
-
Find the difference quotient, \(\frac{f(x+h)-f(x)}{h}\), for the functions given in Problems 39-44. \(f(x)=5 x^{3}\)
-
Let \(P(x)\) be the number of prime numbers less than \(x\). Find a. \(P(10)\) b. \(P(-10)\) c. \(P(100)\)
-
What is your own best learning style? Do you learn best through images? Printed words? Aural sources? How might this approach to learning suit you as you enter a career as a technical communicator?
-
Find the difference quotient, \(\frac{f(x+h)-f(x)}{h}\), for the functions given in Problems 39-44. \(f(x)=x^{3}\)
-
Huan Yue is wondering why the debt to total assets and interest coverage ratios are calculated. Answer her question and explain why the debt to total assets ratio should never be interpreted without...
-
Write a paper detailing a geographic information system (GIS) of your own design that would utilize data in an original manner.
-
Propose a curved-arrow mechanism for the following reaction. Explain why the equilibrium lies to the right. Ph Ph CH toluenesulfonic acidCH
-
Why is trityl chloride much more reactive than the other alkyl halides in Table 17.2? TABLE 17 2 comparison of S,1 Solvolysis Rates of Benzylic and Nonbenzylic Alkyl Halides 25C R-CIHo l + H2O -OH +...
-
What product(s) are expected when each of the following compounds reacts with one equivalent of NBS in CC14 in the presence of light and peroxides? Explain your answers. (a) cyclohexene (b)...
-
sked byChina699 BEMIDJI STATE UNIVERSITY Department of Technology, Art & Design TADT 3217 : Materials Science & Metallurgy Hardness Testing [A continued look at the Heat Treatment of Steel] ...
-
1. Advocate Aurora Sheboygan Memorial Hospital health care product or service to be marketed in your community. 2. Conduct appropriate market research in your community to determine the demographics...
-
How do ethical leaders integrate ethical considerations into strategic decision-making processes, balancing short-term business objectives with long-term ethical imperatives to ensure sustainable...

Study smarter with the SolutionInn App


