Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that
Question:
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that do.
a. CHF=CHF
b. F2C=CH2
c. CH2=CH-CH2-CH3
d.
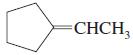
e.
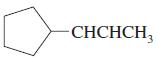
f.
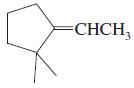
Transcribed Image Text:
CHCH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
The cis trans isomerism is shown by the alkenes only The essenti...View the full answer

Answered By

Mr. VISHAL
I'm pursuing msc. Chemistry (organic specialisation). I have deep knowledge of chemistry and i can explain the concepts in a very simple way. I also like to give lectures on specific topics.
My linkedin profile is:
www.linkedin.com/in/vishal-chnaliya-66a85514b
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star (*) any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may...
-
Which of the following compounds show only a single peak in their 1H NMR spectrum? a. CH3CH2OCH2CH3 b. c. CH,CH,CCI
-
Which of the following compounds are aromatic? a. b. c. Cycloheptatrienyl cation d. e. f. g. Cyclononatetraenyl anion h. CH2=CHCH=CHCH=CH2
-
In Exercises find the positive values of p for which the series converges. n=1 n
-
BH+ ClO-4 is a salt formed from the base B (Kb = 1.00 10-4) and perchloric acid. It dissociates into BH+, a weak acid, and ClO-4, which is neither an acid nor a base. Find the pH of 0.100 M BH+ClO-4.
-
What is the output of the following application? A. 3 B. 9 C. 93 D. The code does not compile because of line g1. E. The code does not compile because of line g2. F. None of the above. package dnd;...
-
Laura Neilson, a job specialist with Vocational and Educational Services for Individuals with Disabilities, responded to an advertisement in a New York newspaper for a position as a shower, a job...
-
During the course of the audit of Nature Sporting Goods for the year ended December 31, 2007, the auditor discovered the following: The accounts receivable confirmation work revealed one pricing...
-
G is an individual GST registrant that files GST returns annually. At the beginning of year 20XX, G incorporated their sole proprietorship into G Inc., a wholly owned subsidiary of G, and registered...
-
Curve Fitting with Constraints. A banks economist has been interested in developing a production function for the bank. The model recognizes three explanatory variables, which are measures of...
-
Two compounds with the formula CH 3 -CH=N-CH 3 are known. (a) Draw a Lewis structure for this molecule, and label the hybridization of each carbon and nitrogen atom. (b) What two compounds have this...
-
Give the relationship between the following pairs of structures. The possible relationships are: same compound cis-trans isomers constitutional isomers (structural isomers) not isomers (different...
-
Bryce, a bank official, is 40 years old, unmarried and has no dependents. During 2022 he engages in the following activities and transactions: a. Being an avid fisherman, Bryce develops an expertise...
-
Sam the CAM quit and his records were corrupted. All we could salvage were the following bits of information: Budget at Completion (BAC) = $120,000 Cumulative Earned Value (EV) = $40,000 Cost...
-
A taxpayer has calculated most of their return, and realizes that they missed on claiming a deduction for their self-employment tax paid. They are in the 22% bracket, and their total calculated...
-
Directions: Write 4 to 5 sentences for each questions What is the expected value of the licensing arrangement to LAB? Assume a 5% royalty fee on any cash flows that Merck receives from Davanrik after...
-
A taxpayer has a vehicle and wishes to depreciate it. They drove 15,000 miles in total during the year, of which 9,250 were business miles and 1,250 were commuting miles. What ratio is used to...
-
You believe you will spend $31,000 a year for 11 years once you retire in 22 years. If the interest rate is 7% per year, how much must you save each year until retirement to meet your retirement...
-
A particle of charge +12 C and mass 3.8 10-5 kg is released from rest in a region where there is a constant electric field of +480 N/C. What is the displacement of the particle after a time of 1.6 ...
-
Find the center of mass of a thin triangular plate bounded by the y-axis and the lines y = x and y = 2 - x if (x, y) = 6x + 3y + 3.
-
What products are formed when gach of the following ethers reacts with concenffated aqueous HI? 2-ethoxy-2,3-dimethylbutane
-
From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each is racemic.) C,H OH/H,O CH CH2 sodium azide
-
Suggest a Williamron other cynthosis, if one is possible, for each of the following compounds. If no Williamson ether synthesis is possible, explain why. (CH3)2CH---S---CH3
-
Year 1 total cash dividends Year 2 total cash dividends Year 3 total cash dividends Year 4 total cash dividends $ 18,000 28,000 300,000 450,000 York's outstanding stock consists of 80,000 shares of...
-
Due to erratic sales of its sole product-a high-capacity battery for laptop computers-PEM, Incorporated, has been experiencing financial difficulty for some time. The company's contribution format...
-
Inventory turnover and number of days' sales in inventory Financial statement data for years ending December 31 for Tango Company follow: 20Y7 20Y6 Cost of goods sold $3,864,000 $4,001,500...

Study smarter with the SolutionInn App


