Consider the following polymer: From which two monomers is the polymer made? OCH,CH,OCCH,CH,COCH,CH,OCCH,CH,C
Question:
Consider the following polymer:
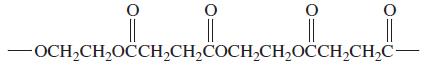
From which two monomers is the polymer made?
Transcribed Image Text:
OCH,CH,OCCH,CH,COCH,CH,OCCH,CH,C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following monomers might you expect to lead to a conducting polymer? a. CH3CH==CHCH3 b. CH CC CH
-
Consider the structure of the following polymer. (a) Draw the monomers you would use to prepare this polymer. (b) Determine whether this polymer is a step-growth polymer or a chain-growth polymer....
-
Consider the following polymer: Is this polymer a homopolymer or a copolymer, and is it formed by addition polymerization or condensation po-lymerization? What is (are) the monomer(s) for this...
-
How much momentum does a car of mass 1 5 0 2 KG have a travels at a consistent speed of 1 4 . 3 m / s?
-
Loss of calcium is a serious problem for older women. To investigate the amount of loss, a researcher measured the initial amount of bone mineral content in the radius bone of the dominant hand of...
-
One companys bottles of grapefruit juice are filled by a machine that is set to dispense an average of 180 milliliters (ml) of liquid. The company has been getting negative feedback from customers...
-
Graph the expectation function for the logistic growth model (12.34) for \(\theta_{1}=10, \theta_{2}=2\), and values of \(\theta_{3}=0.25,1,2,3\), respectively. Overlay these plots on the same set of...
-
The Coca-Cola Company is a global soft drink beverage company (ticker symbol = KO) that is a primary and direct competitor with PepsiCo. The data in Exhibits 12.1312.15 include the actual amounts for...
-
A company has 5 3 6 4 2 3 8 shares outstanding and the stock sells at $ 8 4 . 6 4 per share. What is the market value of equity?
-
Mr. T makes a good recovery and is discharged from the hospital, but a few weeks later you meet him again in the outpatient clinic complaining of headache, blurred vision, and pinkeye. His eyes are...
-
Poly(vinyl chloride) (PVC) is an addition polymer of vinyl chloride, CH 2 = CHCl. Write the equation for the formation of the polymer.
-
Consider the following polymer: From which two monomers is the polymer made? -C(CH2)10NH(CH2)6 NHC(CH2)10CNH(CH2)6NH-
-
Multiple Choice Questions 1. Personal income tax revenue a. 6 only b. 2 only c. 4 only d. 2 and 4 2. Accrued postemployment benefit liability, governmental fund employees a. I and 3 b. 1 only c. 3...
-
Implement a static generic method PairUtil.swap whose argument is a Pair object, using the generic class declared in Section 18.2. The method should return a new pair, with the first and second...
-
Describe how customers and companies are becoming interconnected.
-
Consider the problem of finding the least expensive routes to all cities in a network from a given starting point. For example, in the network shown on the mapbelow, the least expensive route from...
-
Add an instance variable currentSize to our implementation of the LinkedList class. Modify the add, addLast, and remove methods of both the linked list and the list iterator to update the currentSize...
-
Consider the following tree. In which order are the nodes printed by the Binary- SearchTree.print method? The numbers identify the nodes. The data stored in the nodes is not shown. 1 8 6 10
-
Ramon had AGI of $180,000 in 2015. He is considering making a charitable contribution this year to the American Heart Association, a qualified charitable organization. Determine the current allowable...
-
H Corporation has a bond outstanding. It has a coupon rate of 8 percent and a $1000 par value. The bond has 6 years left to maturity but could be called after three years for $1000 plus a call...
-
In a study of formaldehyde oxidation over Ag catalysts, the power rate law over Ag powder was r = kPo2 and over supported Ag it was r = kP0:3O2 P0:3H2CO [62]. The following sequence of steps was...
-
A 250 L rigid tank contains methane at 500 K, 1500 kPa. It is now cooled down to 300 K. Find the mass of methane and the heat transfer using a) ideal gas and b) the methane tables.
-
A rigid insulated tank is separated into two rooms by a stiff plate. Room A of 0.5 m3 contains air at 250 kPa, 300 K and room B of 1 m3 has air at 150 kPa, 1000 K. The plate is removed and the air...
-
A bank can either invest money for three months at 4.00% or for nine months at 4.50%. Ignoring actual/360 day count adjustments for the purpose of this question, the three against nine FRA quote the...
-
Shuggy Otis, an executive at Slapfish Corp. (SC) intends to retire in 11 years. SC just announced that it will start depositing $500.00 at the end of each quarter into each of its workers' retirement...
-
On January 1, the Hanover Beverage Company replaced the palletizing machine on one of its juice lines. The cost of the machine was $195,000. The machine's expected life is five years or 480,000...

Study smarter with the SolutionInn App



