Consider the following reaction: This reaction has been determined to be second order. (a) What is the
Question:
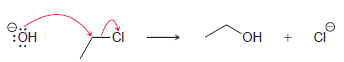
This reaction has been determined to be second order.
(a) What is the rate equation for this reaction?
(b) How will the rate be affected if the concentration of hydroxide is tripled?
(c) How will the rate be affected if the concentration of chloroethane is tripled?
(d) How will the rate be affected if the temperature is raised by 40°C?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





