For each of the following reactions determine whether ÎS for the reaction (ÎS sys ) will be
Question:
a.
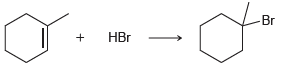
b.
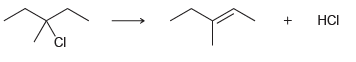
c.
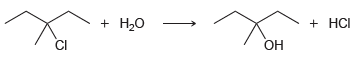
d.
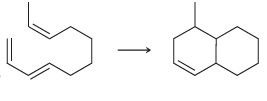
e.
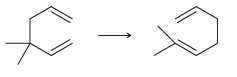
Transcribed Image Text:
Br НBr HCI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
a S sys is expected to be negative a decrease in entropy because ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) For each of the following reactions, predict the sign of ÎH° and ÎS° and discuss briefly how these factors determine the magnitude of K. (b) Based on your general chemical...
-
For each of the following reactions, give the curved-arrow notation and write the analogous Bonsted acid-base reaction. H,C-CH,-Br: + :C=N: H,C-CH-C=N: + :Br:
-
For each of the following reactions. provide the following information. (a) Give the structures of all products (including stereoisomers). (b) If more than one product is formed, give the...
-
You are looking at buying a piece of real estate and you intend to borrow as much as you possibly can from a bank to buy the property. The bank you are dealing with has a requirement that the LVR for...
-
Suppose you want to perform an experiment to verify the problems that can be caused by random insert/remove pairs. Here is a strategy that is not perfectly random, but close enough. You build a tree...
-
Discuss the sampling strategy and technique used to access the appropriate sample. Develop a hypothetical research scenario that would necessitate the use of a Regression-Discontinuity Posttest-Only...
-
Consider a family of call options on a non-dividend-paying stock, each option being identical except for its strike price. The value of the call with strike price \(K\) is denoted by \(C(K)\). Prove...
-
One item is omitted in each of the following summaries of balance sheet and income statement data for four different corporations, East, North, South, and West. Determine the missing amounts,...
-
What challenges might organizations encounter during the implementation and sustenance of Lean Management practices, and what strategies can be employed to overcome these obstacles and ensure...
-
A molded plastic product (p = 1200 kg/m 3 c = 1500 J/kg K, k = 0.30 W/m K) is cooled by exposing one surface to an array of air jets, while the opposite surface is well insulated. The product may...
-
Explain: The United States can make certain toys with greater productive efficiency than can China. Yet we import these toys from China. Relate your answer to the ideas of Adam Smith and David...
-
The percentage that casinos make on the average bet is called the a. Vig. b. Rip. c. Take. d. Rob.
-
What is the gross margin ratio?
-
What is the "crisis" that Rural Alaska is facing over energy cost and generation? How does this crisis impact residents in Rural Alaska? How does it impact the economy of Rural Alaska?
-
What object will the following code produce? glBegin(GL_POLYGON); glColor3f(1, e, e); glVertex3f(-0.6, 0.75, 0.5); glColor3f(0, 1, e); glVertex3f(0.6, -0.75, 0); glColor3f(e, e, 1); glVertex3f(0,...
-
1 ) Night Shades, Inc. manufactures biotech sunglasses. The variable materials cost is $ 1 1 . 1 3 per unit, and the variable labor cost is $ 7 . 2 9 per unit. A ) What is the variable cost per unit?...
-
1 . Prepare a PowerPoint sales presentation that addresses the following: a . Give five main reasons for implementing business analytics in a client's business operations. b . Select three...
-
watched youtube videos and work on authentic leadership and servant leadership. https://youtu.be/ywX3V69EbA8 https://youtu.be/zrxHxGwVEMo Authentic Leadership Servant Leadership
-
Use Fleurys algorithm to determine an Euler circuit. B
-
A copper wire (density = 8.96 g/cm 3 ) has a diameter of 0.25 mm. If a sample of this copper wire has a mass of 22 g, how long is the wire?
-
Which of the following alkyl halides would you expect to undergo Friedel-Crafts reaction without rearrangement? Explain. (a) CH3CH2Cl (b) CH3CH2CH (Cl) CH3 (c) CH3CH2CH2Cl (d) (CH3) CCH2Cl (e)...
-
What is the major mono-substitution product from the Friedel-Crafts reaction of benzene with 1-chloro-2-methylpropane in the presence of AlCl3?
-
Identify the carboxylic acid chloride that might be used in a Friedel-Crafts acylation reaction to prepare each of the followingacylbenzenes: (b) (a)
-
determine how you could check the numbers in the financial report are correct for cash at bank?
-
How and when are allowances formalized into the contract? NOTE: As time went on, the contractor used creative accounting and charged all sorts of marginal costs against the allowances. Is this legal...
-
During the current fiscal year, a company made the following expenditures: Spent $64,928 to overhaul a machine. The overhaul enabled the machine to produce more products per hour than it previously...

Study smarter with the SolutionInn App


