Give the IUPAC name of each of the following compounds. a. CH 3 C = CCH 3
Question:
Give the IUPAC name of each of the following compounds.
a. CH3C = CCH3
b.
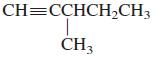
Transcribed Image Text:
CH=CCHCH2CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a CH 3 C CCH 3 As the two Carbons which are Connec...View the full answer

Answered By

Himanshu Kumar
I have worked for various online sites as a doubt solver and subject experts .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the IUPAC name of each of the following compounds. a. b. c. d. CH3CHCH2COOH CH3 CH CHCH CH3 O CH CH CHCCH CH CH2 CH CH3
-
Give the IUPAC name of each of the following compounds. a. b. c. d. COOH CH,CCH CH3 CH3 CH3 CH,CCOCH3 CH2CH3
-
Give the IUPAC name for each of the following compounds. a. b. CH C-CHCHCH CH CH2 CH3 CH CH2CH2CHCH2CH2CH3 CH CH2
-
1. Consider a local department store which only sells jeans (J) and sweaters (S). Jeans cost $20 and sweaters cost $30. For each of the examples below, graph the budget constraint our consumer faces,...
-
For the data of Exercise 2.57, calculate s2 by using the alternative formula. In Exercise 2.57 3 8.
-
Consider the system of Problem 10.1. For the same values of \(m\) and \(T\), find its output \(c(t)\) in conventional BPF domain using the convolution matrix. Finally, compare the convolution result...
-
A used car dealer sold an automobile to a customer (Robert) on an installment basis. The installment sales contract calls for Robert to make monthly payments of principal and interest. The contract...
-
Last year Clark Company issued a 10-year, 12% semiannual coupon bond at its par value of $1,000. Currently, the bond can be called in 4 years at a price of $1,060 and it sells for $1,100. a. What are...
-
16. 17. The IUPAC nomenclature of an element with electronic configuration [Rn] 5f46d17s is: (a) Unnibium (b) Unnilunium (c) Unnilquandium (d) Unniltrium The compound(s) that is (are) removed as slag...
-
The box plot below shows the amount spent for books and supplies per year by students at four year public colleges. a. Estimate the median amount spent. b. Estimate the first and third quartiles for...
-
Give the IUPAC names and include the cis or trans label for each of the isomers of CH 3 CH = CHCH 3 .
-
Write the IUPAC name of each of the following hydrocarbons. a. b. b. CH 3 C = CCH 2 CH 3 CH3CHC=CH CH3
-
Differentiate. G(x) = x 2 - 2/2x + 1
-
A new 15-year mortgage had an initial balance of $123.8 thousand and an interest rate of 5.9%. What is the balance of this mortgage after 57 months? Assume that the borrower has made only the...
-
Stoney Corporation invest $6,500 each in Projects X and Y. Cash flows from these investment are listed below Investment X Investment Y Year 1 $1000 $1300 Year 2 1800 2000 Year 3 1700 1100 Year 4 2000...
-
Jerry is a delivery person for Pretty Flowers Florists. His primary job responsibility is to deliver floral arrangements in the companys van. While driving a floral arrangement to a wedding, Jerry...
-
Bank A has a loan with firm X for $15.0 million. The loan is due in full at the end of year 3. To protect itself, the bank takes a credit default swap with LowRisk Insurance Company at a cost of 2.5%...
-
Walk me through the steps of setting up an effective incentive plan for the following situation. Situation: You are a manager at a large tech company, and notice that your employees consistently turn...
-
A sodium atom (Z = 11) contains 11 protons in its nucleus. Strictly speaking, the Bohr model does not apply, because the neutral atom contains 11 electrons instead of a single electron. However, we...
-
TRUE OR FALSE: 1. Banks with a significantly large share of fixed-interest rate home loans are less exposed to interest rate risks. 2. Although Australian banks are pretty big, they are not...
-
10 kg of water in a piston cylinder arrangement exists as saturated liquid/vapor at 100 kPa, with a quality of 50%. It is now heated so the volume triples. The mass of the piston is such that a...
-
Two tanks are connected by a valve and line as shown in Fig. P5.62. The volumes are both 1 m3 with R-134a at 20C, quality 15% in A and tank B is evacuated. The valve is opened and saturated vapor...
-
Consider the same system as in the previous problem. Let the valve be opened and transfer enough heat to both tanks so all the liquid disappears. Find the necessary heat transfer.
-
Consider the following information: State Probability ABC Inc.'s Return Boom 0.25 15% Normal 0.50 8% Slowdown 0.15 4% Recession 0.10 -3% i. 11. iii. What is the expected return? What is the variance?...
-
The mythical country of Quitar has just established a policy to give very generous in-kind benefits to the nation's poor, currently defined as those earning less than $10,000 a year. Several years...
-
Question 1 The external loadings imposed on a horizontal beam is shown in Figure Q1(a). The beam is pivoted at Point A. (a) (b) 100 N/m 50 N/m 20 N 40 cm 50 cm Figure Q1 70 cm Apply the principle of...

Study smarter with the SolutionInn App


