In Chapter 10, we will see that an acetylide ion (formed by treatment of acetylene with a
Question:
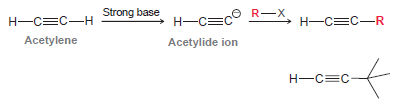
This reaction provides a useful method for making a variety of substituted alkynes. Determine whether this process can be used to make the following alkyne. Explain your answer.
Transcribed Image Text:
Strong base O R-X Н—С—С Н—СС—R Н—СС-н Acetylene Acetylide ion Н—с—С
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
No Preparation of this compo...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Can a buffer be made by combining a strong acid with a strong base? Why or why not?
-
A large slab of concrete, 5 8 0.3 m is used as a thermal storage mass in a solar-heated house. If the slab cools overnight from 23C to 18C in an 18C house, what is the net entropy change associated...
-
In the following acid-base reactions, 1. Determine which species are acting as electrophiles (acids) and which are acting as nucleophiles (bases). 2. Use the curved-arrow formalism to show the...
-
A car costs 12,000. It will be kept for three years, and then sold for 3,000. Calculate the depreciation for each year using (a) the reducing balance method, using a depreciation rate of 35 per cent,...
-
What issues confront the company as of mid-2012? What should Starbuck's management be worried about?
-
Prove that model (7.15) is indeed the model for the row-effect association. log (ij) = ++iy, 1iI,1 j J. (7.15)
-
Write requirements for the following products and services: a. Computer. b. Airplane. c. Food Processor. d. Online Banking e. Life Insurance. f. Party Planning.
-
Five years ago, Brian and his brother Boyd formed Stewart Corp., a golf apparel manufacturing corporation. At that time, Brian contributed $300,000 to the corporation in exchange for 50% of its...
-
Use the DEFINITION to prove the following: (n + 4) O(n + 4n) E (n + 2n) (n + 2) E
-
1. Snyders of Hanover, which sells about 80 million bags of pretzels, snack chips, and organic snack items each year, had its financial department use spreadsheets and manual processes for much of...
-
Identify the configuration of each chirality center in the following compounds: a. b. c. d. e. f. g. h. i. Et OH Me NH2
-
Predict the product(s) obtained when each of the following compounds is treated with chloromethane and aluminum trichloride. Some of the compounds might be unreactive. For those that are reactive,...
-
Gerardo Rogers is conducting a review of the payroll files for each employee at Meejo Games. Which of the following items must be present in the file? 1. Basis upon which compensation is paid 2....
-
Suppose that no amount of other goods can compensate for a loss in health. How would the individuals indifference curves look? Is this a reasonable assumption in terms of what we actually see taking...
-
A company starts in business on 1 January 2011. You are to write up the vans account and the provision for depreciation account for the year ended 31 December 2011 from the information given below....
-
T. Penketh is an accountant with a salary of 2,000 per month plus bonus, which for May 2012 was 400. He pays superannuation contributions of 5 per cent of gross pay, and these are allowed as reliefs...
-
A business started trading on 1 January 2010. During the two years ended 31 December 2010 and 2011 the following debts were written off to the Bad Debts Account on the dates stated: On 31 December...
-
Someone says the following: Lifestyle may be the most important determinant of health status, but changing lifestyles may not be the least costly way to improve population health status. Explain the...
-
Derive the output equations for the asynchronous sequential circuit shown in Figure P5.34. Also, determine the state table and flow table. X a a Do Do Da Da Do Dop A B
-
Write the general quadratic equation y2 - 8y - 4x + 28 = 0 in standard form. Determine the vertex, focus, and directrix of the parabola defined by this equation. Sketch a graph.
-
The Wilkinson catalyst chlorotris (triphenylphosphine) rhodium(I), ClRh(PPh0)3, brings about the catalytic hydrogenation of an alkene in homogeneous solution: (a) Using the following mechanistic...
-
Characterize each step of the mechanism in Eq. 18.42b in terms of the fundamental processes discussed in the previous section. Give the electron count and the oxidation state of the metal in each...
-
Characterize each step of the mechanism in Eq. 18.42b in terms of the fundamental processes discussed in the previous section. Give the electron count and the oxidation state of the metal in each...
-
1. Define latent heat and how it is different than specific heat capacity. 2. Describe how a phase diagram changes when changing from a solid to a liquid. 3. Describe how work done is related to a...
-
In a large vaccination clinic, patients arrive at the rate of 50 per hour. The clinic is staffed with five nurses and it takes on average 6 minutes for a nurse to vaccinate a patient. Both patient...
-
Calculate the missing value. Beginning cash balance add : cash receipts Collection of notes receivable Proceeds from sale of securities collection from credit sales Total receipts Total available...

Study smarter with the SolutionInn App


