Lactones can be prepared from diethyl malonate and epoxides. Diethyl malonate is treated with a base, followed
Question:
![1) NaOEt EtO EtO OEt 2) OEt Н,о Heat [H,O*] Н,о* Heat Но Но ОН (-CO2) ОН ОН](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1525/0/6/8/3845ae6b260351221525068355373.jpg)
Using this process, identify what reagents you would need to prepare the following compound:
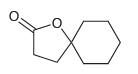
Transcribed Image Text:
1) NaOEt EtO EtO OEt 2) OEt Н,о Heat [H,O*] Н,о* Heat Но Но ОН (-CO2) ОН ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Eto OE...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline syntheses of each of the following from diethyl malonate and any other required reagents: (a) 2-Methylbutanoic acid (b) 4-Methyl-1-pentanol (c) (d) OH
-
Thiols can be prepared from the reaction of thiourea with an alkyl halide, followed by hydroxide-ion-promoted hydrolysis. a. Propose a mechanism for the reaction. b. What thiol would be formed if the...
-
When ethylene oxide is treated with a strong nucleophile, the epoxide ring is opened to form an alkoxide ion that can function as a nucleophile to attack another molecule of ethylene oxide. This...
-
aj b) If the magnitude of the force F is 700 N, what is the value of the internal moment at point A in N.m? d) 0.5 m 540 180 210 A 0.8 m 240 Bo birak F A
-
For the composite wall idealized by the one-dimensional model shown in Figure P13-7, determine the interface temperatures. For element 1, let Kxx = 5 W (m ( oC) for element 2, Kxx = 10 W (m ( oC);...
-
The current spot price of one GBP is \( 1\).2. The continuously compounded interest rate in the Eurozone is \(2.4 \%\), whereas the corresponding UK interest rate is \(3.1 \%\). If the forward price...
-
Great Western Bank has offered a special certificate of deposit (CD) tied to the S\&P 500. Funds are deposited into the account at the beginning of a month and are held in the account for 3 years....
-
Walden Green provides custom farming services to owners of 5-acre wheat fields. In July, he earned $2,400 by cutting, turning, and balling 3,000 bales. In the same month, the incurred the following...
-
If today's 1-year interest rate is 5%, and you expect 1-year interest rates to be 6% next year and 6.25% the year after that, compute the term structure and draw today's yield curve based on the...
-
Suppose that it costs a firm $2 to produce each unit of its output. What is its supply function? Graph it.
-
Draw the structure of the product with molecular formula C 10 H 10 O that is obtained when the compound below is heated with aqueous acid. CN CN C10H100 Heat
-
Predict the major product of the following transformation. CO2ET C10H100 Heat
-
Find the exact interest on $1,000 for 60 days at 5.3% annual interest rate.
-
Write a list of key points to be made in a recommendation to the board of directors on the implementation of an expenditure control process for capital investment plans.
-
Although in some markets we have moderated our rate of growth in space, overall we have chosen to sustain strong growth in selling area and this will continue. With reductions in site, build and...
-
Explain how a project having a positive net present value will also have a profitability index greater than 1.0.
-
A company is considering investing $100,000 in a new machine that will reduce its annual cash operating costs as follows: Calculate the payback period to the nearest 0.1 years. Year 1234 Operating...
-
Use your calculator to check the discount factors for the present value of 1 at the end of one year, two years and three years for a discount rate of 10%. Write a parallel table for 8 per cent and 12...
-
Ms. French is a successful attorney in the 37 percent marginal tax bracket. During the past several years, she provided legal services to her great-uncle, who is 78 years old and in failing health....
-
Use translations to graph f. f(x) = x-/2 +1
-
Outline a synthesis for each of the following compounds from the indicated starting material and any other reagents. 4-methyl-3-nitropyridine from - lpicoline
-
Predict the predominant product in each of the following reactions. Explain your answer. 3,4-dibromopyridine + NH3, heat (C5H5BrN2)
-
Using bases (B:) and acids (+BH) as needed, provide a curved-arrow mechanism for the conversion of the c-amino acid serine into formaldehyde and glycine (Eq. 25.53, p. 1242). Eq. 25.53 formaldehyde...
-
Suppose the correlation between the stock euro returns of Siemens and the USD/EUR exchange rate is 0.2. The standard deviation of the USD/EUR is 10% and the standard deviation of Siemens's stock euro...
-
list and describe the three key client-related factors that the advisor is required to consider when developing a "suitable" investment portfolio for their client. Please cite resources used
-
Year 1 2 3 Amount ($) 2000 3000 4000 An investment made today will pays you the above cash flows at the end of each year. If your required rate of return is 5% annual interest, how much will you pay...

Study smarter with the SolutionInn App


