Name all of the functional groups present in the following molecule. CH H-CHCH CH3
Question:
Name all of the functional groups present in the following molecule.
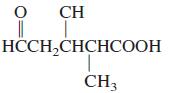
Transcribed Image Text:
CH НССH-CHCHСООН CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
In this molecule two functional groups are ...View the full answer

Answered By

K sujatha
I am chemistry expert I have expertise in different streams of chemistry like physical chemistry , organic chemistry and inorganic chemistry
Chemistry is the subject which is related to life process and daily life activities . so this application leads to me choose my career in chemistry .
My first experience is working with chaithanya institutions which are very famous and I hanadled sixth children and explained foundation concepts for 2 years
next my journey started with Narayana in that i handled high school section their i got so much knowledge in subject and experince , hard work teaches everything . so in this institution i worked nearly 5 years
Enhanced my subject knowledge and improved myself to give easy of ways learning
Teaching is art so i should make it very easily
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify all of the functional groups in the following molecule. OH HOO
-
Identify' the functional groups present in the following compounds. a. b. c. OH CH, I CH3 Testosterone CH3O HO CH Vanillin C-OCH H2N CH-C NH CHCH2 CHC-OH Aspartame
-
Identify all of the functional groups in each of the following compounds: (a) (b) (c) (d) (e) (f) (g) Vitamin D3 HO OMe Aspartame O NH2 NH2 Amphetamine Me Cholesterol HO OCH2CH3 Demerol CH A...
-
Which statement about the Java variable and literal is NOT true? a. A global variablewill be initalised by the complier automatically. b. Literals can also be used as the lvalue in an express. c. An...
-
Using the data on testosterone x4 for male alligators: (a) Make separate dot plots for the Lake Apopka and Lake Woodruff alligators. (b) Calculate the sample means for each group. (c) Do the...
-
Nationally, the proportion of red cars on the road is 0.12. A statistically minded fan of the Philadelphia Phillies (whose team color is red) wonders if Phillies fans are more likely to drive red...
-
Suppose that the full model is \(y_{i}=\beta_{0}+\beta_{1} x_{i 1}+\beta_{2} x_{i 2}+\varepsilon_{i}, i=1,2, \ldots, n\), where \(x_{i 1}\) and \(x_{i 2}\) have been coded so that...
-
For each of the mixed streams of cash flows shown in the following table, determine the future value at the end of the final year if deposits are made into an account paying annual interest of 12%,...
-
The second capital budgeting decision which you were asked to analyze involves choosing between two mutually exclusive projects, S and L, whose cash flows are set forth below: Year 012345 Expected...
-
Based on the three tables and the attributes below, answer the question for Requirement A and write SQL commands for Requirements B to E to retrieve the data from the database. Salesltems Sales...
-
For following molecular model, where C atoms are black and H atoms are light blue: a. Write the molecular formula. b. Write the condensed structural formula.
-
Draw the condensed structural formulas for a primary, a secondary, and a tertiary alcohol, each containing five carbon atoms.
-
The Executive Board of the Mens Professional Tennis Tour (MPTT) has announced changes to its rules pertaining to doubles play for the upcoming tour season. The Executive Board consists of three...
-
Give a SQL query that lists all products in the invoice database of Section 24.2. Invoice Invoice_ Number INTEGER 11731 11732 11733 Customer_ Number INTEGER 3175 3176 3175 Payment DECIMAL (10, 2) 0...
-
Add a method void depthFirst(Visitor v) to the Tree class of Section 17.4. Keep visiting until the visit method returns false.
-
What is the difference between a thread that sleeps by calling sleep and a thread that waits by calling await?
-
Write a program that sorts an array of bank accounts by increasing balance. Pass an appropriate lambda expression to Arrays.sort.
-
Playfair cipher. Another way of thwarting a simple letter frequency analysis of an encrypted text is to encrypt pairs of letters together. A simple scheme to do this is the Playfair cipher. You pick...
-
A number of years ago, Lee acquired a 20% interest in the BlueSky Partnership for $60,000. The partnership was profitable through 2014, and Lee's amount at risk in the partnership interest was...
-
Tiger, Inc. signed a lease for equipment on July 1, 2007.The lease is for 10 years (the useful life of the asset).The first of 10 equal annual payments of $500,000 was made on July 1, 2007.The...
-
Use the ideal gas air table A.7 to evaluate the heat capacity Cp at 300 K as a slope of the curve h(T) by h/T. How much larger is it at 1000 K and 1500 K.
-
We want to find the change in u for carbon dioxide between 600 K and 1200 K. a) Find it from a constant Cvo from table A.5 b) Find it from a Cvo evaluated from equation in A.6 at the average T. c)...
-
We want to find the change in u for oxygen gas between 600 K and 1200 K. a) Find it from a constant Cvo from table A.5 b) Find it from a Cvo evaluated from equation in A.6 at the average T. c) Find...
-
Janice will need to pay $200 at the end of every month for the next 12 months, except for the payment of the 8th month. What is the present value, assuming a rate of 4%, compounded quarterly?
-
List and explain two management tools in the planning process and two measurable performance indicators. Explain in detail.
-
What does a high PE tell us about the value of the stock price (over or under valued)? What does a low PE tell us about the value of the stock price (over or under valued)? Be specific with your...

Study smarter with the SolutionInn App


