Predict the major product(s) for each of the following reactions: a. b. c. d. (PPH3)3RHCI H;o*
Question:
a.
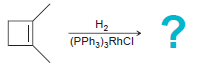
b.
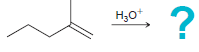
c.
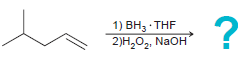
d.
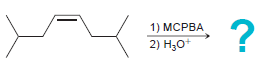
Transcribed Image Text:
На На (PPH3)3RHCI H;o*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a b ...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product expected for each of the following reactions: (a) (b) (c) (d) (e) (f) ? XS HCI CI 1) xs NANH/NH3 2) H,0 CI
-
Predict the major product obtained when each of the following compounds is treated with Birch conditions. a. b. c. d. e. f. OH
-
Predict the major product obtained when each of the following compounds is treated with bromine (Br 2 ) together with sodium hydroxide (NaOH) followed by aqueous acid (H 3 O + ). (a) (b) (c)
-
GoFast Corp. wishes to renovate the property so that he can sell it at a higher price.The after-tax cash flow over next year up to RM320,000 if the property was renovated.The renovation cost is...
-
Give an algorithm that finds the strongly connected components in only one depth-first search. Use an algorithm similar to the biconnectivity algorithm.
-
Describe two conflicts of interest that occur in universal banks.
-
Discuss the objectives and classification of criminal offenses.
-
Shanahan Corporation produces three types of media: CDs, DVDs, and double-layer DVDs. Shanahan purchases cases for the media from a firm in Mexico and purchases the labels from another supplier in...
-
David Ltd sold the following machine in 2017: Cost Purchase date Useful life Residual value Selling date $150,000 1 July 2014 5 years $10,000 1 July 2017 $74,000 Sales proceeds Required: 1. Prepare...
-
The Economics Principles Task (EPT) is worth 30% of the final mark. The EPT is a 'take-home' exercise with a set of tasks to do. The EPT is based on lectures/modules: El (M6), E2 (M7) and E3 (M8)....
-
Paclitaxel (marketed under the trade name TaxolTM) is found in the bark of the Pacific yew tree, Taxus berevifolia, and is used in the treatment of cancer: (a) Draw the enantiomer of paclitaxel. (b)...
-
Using any compounds that contain two carbon atoms or fewer, show a way to prepare a racemic mixture of (2R, 3S) - and (2S, 3R) -2, 3-dihydroxypentane.
-
With a classmate, choose a U.S.-based company whose products you think will do well in certain overseas markets. The company can be anything from a music group to a clothing retaileranything that...
-
Why do you think cognitive ability factor has a significant influence on learning ?
-
Hansen bought his primary residence on 1/1/2010 for $400,000. He put $50,000 of his own money as the down payment, and borrowed $350,000 from a bank for the remainder of the purchase price. On...
-
With the following data, determine the statement of retained earnings and market ratios: Statement of retained earnings Initial retained income 790,000 Operating income 1,480,000 Interest expenses...
-
Charter coffee comes in two sizes. A 30.6-ounce costs 7.98. What is the unit price for the 30.6-ounce package? Charter coffee comes in two sizes. A 30.6-ounce costs 7.98. What is the unit price for...
-
Sandy's employer has provided her with $200,000 of group term life insurance. Sandy is 51 and pays $100 annually for her group term life insurance. If the cost per $1,000 of coverage per month is...
-
Determine the winner using the plurality with elimination method. The 12 members of the executive committee of the Student Senate must vote for a student representative for the college board of...
-
An 8.0 kg crate is pulled 5.0 m up a 30 incline by a rope angled 18 above the incline. The tension in the rope is 120 N, and the crates coefficient of kinetic friction on the incline is 0.25. a. How...
-
How many electrons does each of the following elements have in its outermost electron shell? (a) Magnesium (b) Molybdenum (c) Selenium
-
Draw a molecule of chloroform, CHCl3, using solid, wedged, and dashed lines to show its tetrahedral geometry.
-
Convert the following representation of ethane, C2H6. Into a conventional drawing that uses solid, wedged, and dashed lines to indicate tetrahedral geometry around each carbon (gray = c, ivory =H).
-
Milden Company is a merchandiser that plans to sell 4 1 , 0 0 0 units during the next quarter at a selling price of $ 5 9 per unit. The company also gathered the following cost estimates for the next...
-
How do organizational learning theories, such as the learning organization concept and knowledge management practices, contribute to sustained competitive advantage and organizational resilience in...
-
Harbert, Incorporated had a beginning balance of $12,000 in its Accounts Receivable account. The ending balance of Accounts Receivable was $10,500. During the period, Harbert collects $72,000 of its...

Study smarter with the SolutionInn App


