Starting with sodium azide as your source of nitrogen and using any other reagents of your choice,
Question:
Compounds in 23.18
(a)
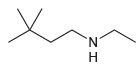
(b)
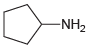
(c)
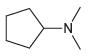
(d)
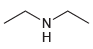
(e)
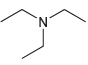
(f)
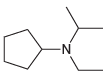
Transcribed Image Text:
-NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The first alkyl group ...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using ammonia as your source of nitrogen, show the reagents you would use to prepare each of the following amines: (a) (b) (c) (d) (e) (f) Z
-
Starting with the following compound and using any other reagents of your choice, outline a synthesis for trimyristin. H.
-
In each of the following indicate which reaction will occur faster. Explain your reasoning. (a) CH3CH2CH2CH2Br or CH3CH2CH2CH2I with sodium cyanide in dimethyl sulfoxide (b) 1-Chloro-2-methylbutane...
-
Rice and Flower were partners sharing profit and loss equally. Statement of Financial Position as at 31 December 2020. Non current Assets Premises Machinery Vehicles Fittings Current Assets Inventory...
-
Using the following data for a hypothetical economy, calculate gross domestic product (GDP), net domestic income (NDI), and government budget surplus (BS):...
-
Joint cost allocation, ending work in process inventories. Tastee Freez, Inc., produces two specialty ice cream mix flavors for soft serve ice cream machines. The two flavors, Extreme Chocolate and...
-
Show that Eq. 9.22 reduces to Eq. 9.8 when the force is constant. Equations WFxAx (constant force exerted on particle, one dimension). (9.8)
-
R&R is a public corporation that, as of December 31, 2009, is subject to a year-end integrated audit by its independent auditing firm, Young & Young. An excerpt of Young & Youngs audit opinion...
-
31 The number of protons, electrons and neutrons in aluminium ion Al+ is Protons A. 27 B. 13 C. ABCD 32 32. D. 13 10 Electron 27 neutrons 14 14 14 10 14 17 14 The formula of the compound formed...
-
A certain amount of mercury is compressed from an initial pressure of 500 MPa to a certain amount of final pressure. The final pressure of mercury required, to reduce a unit volume of mercury by 10%...
-
Draw the structure of each of the following compounds: (a) Cyclohexylmethylamine (b) Tricyclobutylamine (c) 2,4-Diethylaniline (d) (1R,2S)-2-Methylcyclohexanamine (e) ortho-Aminobenzaldehyde
-
What is the difference between a firms mission and its objectives? Why is it important that both are conveyed clearly to employees and to customers?
-
The height H(ft) of a palm tree after growing for t years is given by a. Find the trees height when t = 0, t = 4, and t = 8. b. Find the trees average height for 0 t 8. H = Vt + 1 + 5t1/3 for 0 t ...
-
Now that you have learned the basics of income taxation, employment income or loss, and the taxable income and payable for individuals, let's now discuss the Capital Cost Allowance (CCA) as a...
-
You must access standard recipe for a beef dish and calculate the ingredients needed for the two portions you will prepare and plate. Across the recipes, you must use the following preparation...
-
When would a taxpayer include California Schedule D in their tax return and does CA have special rules for Long Term Capital Gains?
-
On August 19 2022 Kakashi sold stocks directly to buyer. As a result of the transaction, Kakashi paid to the BIR 40,180 representing the total taxes on the transaction. The stocks are not traded in...
-
Question Discuss the Main features and tax advantages of Furnished holiday lettings ?
-
Answer these questions in light of HIPPA requirements: Does HIPAA apply to the RTGM system? Why or why not? How should the system ensure data security during transmission between a patients mobile...
-
True & False The basis of an asset must be reduced by the depreciation allowable, 2. Adjusted gross income (AGI) is the basis for a number of phase-outs of deductions. 3. A change to adjusted gross...
-
An all-suprafacial [3,3] sigma tropic rearrangement could in principle take place through either a chair-like or a boat-like transition statel When the terpene germacrone is distilled under reduced...
-
Explainusing (a) resonance argumeots and (b) molecular orbital arguments why the unpaired electron in the allyl radical is delocalized to carbon-l and carbon-3 but not to carbon-2.
-
Explainusing (a) resonance argumeots and (b) molecular orbital arguments why the unpaired electron in the allyl radical is delocalized to carbon-l and carbon-3 but not to carbon-2.
-
Sweeten Company had no jobs in progress at the beginning of the year and no beginning inventories. It started, completed, and sold only two jobs during the year-Job P and Job Q. The company uses a...
-
1. Review the following tracings, identify the type of BBB, and then explain how you determined your answers 2. Review the following tracing. Do you suspect left ventricular hypertrophy? Explain how...
-
What are signs that its time to cut corners to get the product launched, and what would you cut?

Study smarter with the SolutionInn App


