Using three- and one-letter abbreviations, show the sequence of amino acid residues in the following pentapeptide. H2N
Question:
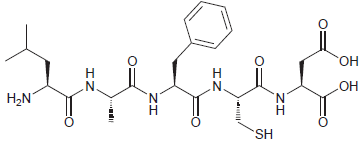
Transcribed Image Text:
Он ОН H2N SH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (12 reviews)
LeuA...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Glucagon is a peptide hormone produced by the pancreas that, with insulin, regulates blood glucose levels. Glucagon is comprised of 29 amino acid residues. Treatment with trypsin yields four...
-
Draw a bond-line structure of the peptide that corresponds with the following sequence of amino acid residues, and identify the N terminus and C terminus: Trp-Val-Ser-Met-Gly-Glu
-
Write structural formulas showing the constitution of each of the following dipeptides. Rewrite each sequence using one-letter abbreviations for the amino acids. (a) Gly-Ala (d) Gly-Glu (b) Ala-Phe...
-
The Whitewater LLP is equally owned by three partners and has the following balance sheet at the end of the current tax year: Partner Petula is an active (i.e., general) partner retiring from the...
-
Ocean Company sells a product with a contribution margin ratio of 80%. Fixed costs are $2,800 per month. What amount of sales (in dollars) must Ocean Company have to break even? If each unit sells...
-
Mike Szabo Company engaged in the following transactions during the month of December: December 2 Made credit sales of $4,000 (accepted accounts receivable). 6 Made cash sales of $2,500. 10 Paid...
-
Creusot-Loire, a French company, was the project engineer for the construction of ammonia plants in former Yugoslavia and Syria. Creusot-Loire contracted with Coppus Engineering for the purchase of...
-
Wyandotte Chemical Company sells various chemicals to the automobile industry. Wyandotte currently sells 30,000 gallons of polyol per year at an average price of $15 per gallon. Fixed costs of...
-
what happens when ou use an aggregation function in a calculated column?
-
St. Josephs Hospital began operations in December 2019 and had patient service revenues totaling $1,290,000(based on customary rates) for the month. Of this, $141,000 is billed to patients,...
-
Draw the structure of each of the following peptides: (a) Leu-Ala-Gly (b) Cys-Asp-Ala-Gly (c) Met-Lys-His-Tyr-Ser-Phe-Val
-
Determine which of the following peptides will have a higher molecular weight. Cys-Tyr-Leu or Cys-Phe-Ile
-
An accounting professor at the University of California at inventory, accounts receivable, and order backlogs. These are the strongest indicators and are more closely related to stock returns than...
-
ABC Corp. expects that a significant portion of its deferred tax assets will not be realized. Which section of the Accounting Standard Codification should ABC consult to determine whether a valuation...
-
What kind of appraisal reports are typically used for income-generating properties?
-
A retail store purchases a specific model of Samsung cell phones from Samsung. A unit of these phones, on average, costs $800 dollars to manufacturer. It is known that the annual demand curve for...
-
Viatris Company currently has a capital structure of 70 percent debt and 30 percent equity, but is considering a new product that will be produced and marketed by a separate division. The new...
-
What are multiple ways a company can hold a competitive advantage over another?
-
Suppose that you have been hired to develop a website-based sales system for a large international retail sales firm. Discuss some environmental issues that are specific to the Web design of your...
-
How does the organizational structure of an MNC influence its strategy implementation?
-
Predict the products you would expect from the reaction of NaBH4 with the following compounds. You may assume that these reactions take place in methanol as the solvent. (a) CH3--(CH2)8--CHO (c) Ph...
-
Repeat Problem 10-24 using LiAlH4 (followed by hydrolysis) as the reagent.
-
Show how you would synthesize the following alcohols by reducing appropriate carbonyl compounds. (a) Heptan-1-ol (b) Heptan-2-ol (c) 2-methylhexan-3-ol (d)
-
What are the key segments in the premium dog market? What key characteristics do customers in the premium dog segment share? How does this influence their buying process for their dogs? Explain the...
-
I have seen an example of an emerging approach that has been successful in the commercial world. Competition from other businesses operating in the same industry was fierce for the company I worked...
-
give me some Marketing strategies for a Trade School to Engage Students How make those strategies works? Also, ideas to Recruit Students for Trade School with Modern Marketing Tactics

Study smarter with the SolutionInn App


