Valine has the structure Draw the zwitterion that would exist at neutral pH. H CH3CH-C-COOH - CH3
Question:
Valine has the structure
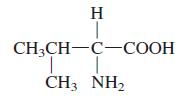
Draw the zwitterion that would exist at neutral pH.
Transcribed Image Text:
H CH3CH-C-COOH -СООН CH3 NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
Zwitterion ...View the full answer

Answered By

Himanshu Kumar
I have worked for various online sites as a doubt solver and subject experts .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Alanine has the structure Draw the zwitterion that would exist at neutral pH. CH3-C-COOH NH
-
Which one of the following five structures is a zwitterion? (a) (b) - O 2 CCH 2 CH 2 CO 2 - (c) H 3 N + CH 2 CO 2 - (d) CH 3 (CH 2 ) 16 CO 2 - K + (e) 0,CCH,CNH,
-
Consider the fully protonated amino acid valine: where the numbers denote the pKa values. (b) Calculate the predominant form of valine at pH 1.0, 7.0, and 12.0. (c) Calculate the isoelectric point of...
-
The top 5 stocks in the S&P 500 index, when ranked by market capitalization, make up 22% of the total market capitalization of the S&P 500 index. Numerical estimates of the mean (or expected) rates...
-
The number of automobile accidents reported per month helps to identify intersections that require improvement. The number of crashes per month reported at an intersection near a university campus in...
-
ACT Doorland was founded in 1957 as a small concern whose core business was the supply and installation of garage doors. Since then it has diversified its interests and expanded to become Canberras...
-
In a train moving due north at \(3.1 \mathrm{~m} / \mathrm{s}\) relative to Earth, a passenger carrying a suitcase walks due north down the aisle at \(1.2 \mathrm{~m} / \mathrm{s}\) relative to the...
-
The sales department of C. Mack Manufacturing Co. has forecast sales for its single product to be 20,000 units for June, with three-quarters of the sales expected in the East region and one fourth in...
-
QUESTION 3 (20 MKS) a) Obtain the time-independent Schrodinger Wave equation from the time dependent equation. [10 mks] b) Solve the time-independent Schrodinger equation given that,(x) = Aekx [10...
-
A consumer group claims that the mean annual consumption of high fructose corn syrup by a person in the United States is 48.8 pounds. A random sample of 120 people in the United States has a mean...
-
Consider the following polymer: From which two monomers is the polymer made? -C(CH2)10NH(CH2)6 NHC(CH2)10CNH(CH2)6NH-
-
If a sample of DNA isolated from a microorganism culture were analyzed and found to contain 1.5 mol of cytosine nucleotides and 0.5 mol of adenosine nucleotides, what would be the amounts of guanine...
-
What problems have accompanied Zuckerberg's initially promising goals for Internet.org? Investigate the current state of what Facebook is doing to curtail its use as a platform to spread violence. Is...
-
What is the kinetic energy of a 1 2 9 . 3 cm thin uniform rod with a mass of 4 7 6 . 1 g that is rotating about its center at 3 . 2 6 rad / s ? Give your answer in Joules.
-
explain why the type of becerage sold is an example of a catwgorical variable.
-
Describe the cost recovery methods for recovering the cost of personal property, real property, intangible assets, and natural resources.
-
On January 1, 2018, Waterway Industries had 116000 shares of its $5 par value common stock outstanding. On June 1, the corporation acquired 9800 shares of stock to be held in the treasury. On...
-
You are in charge of forecasting revenue for the government. Assume the government is going to increase the tax rate. What type of taxpayer is likely to respond to this tax rate increase by working...
-
Find the de Broglie wavelength of an electron with a speed of 0.88c. Take relativistic effects into account.
-
What is a make-or-buy decision?
-
A piston cylinder contains 3 kg of air at 20oC and 300 kPa. It is now heated up in a constant pressure process to 600 K.
-
An insulated cylinder is divided into two parts of 1 m3 each by an initially locked piston. Side A has air at 200 kPa, 300 K, and side B has air at 1.0 MPa, 1000 K. The piston is now unlocked so it...
-
A piston cylinder contains air at 600 kPa, 290 K and a volume of 0.01 m3. A constant pressure process gives 54 kJ of work out. Find the final temperature of the air and the heat transfer input.
-
Randy Sneaker Company is a maker of mid-priced sneakers. They have recently made a new running sneaker but it has not met its sales expectations. As a result, the company president has asked you to...
-
a. Factorize the following expression 6x+13x-5 b. Express 20% as a decimal number c. Simplify (3y+5) (2y-3) - (y+7)(5y - 1). d. Simplify 7x+x-x-5x-3x-6x4 e. Solve -3x+4 <11 hence represent it...
-
Q4(a) Personal protective equipment (PPE) refers to the equipment worn to minimize the worker's exposure to the hazards which causes occupational injuries and illness. Explain three (3) factors which...

Study smarter with the SolutionInn App


