What is the common name of each of the following compounds? a. b. CH3 CH;OCCH3 CH3
Question:
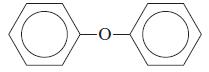
What is the common name of each of the following compounds?
a.
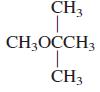
b.
Transcribed Image Text:
CH3 CH;OCCH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What is the Common name of (CH3)2C(Cl)CH(OH)CH2CH3?
-
What is the IUPAC name of each of the following compounds? a. b. c. d. CH3 CH3CH2CHCHCH3 CH3 CH2CH3 CH3CH2CH2CHCH2CH3 CH3 CH3CHCH2CH2CHCH2CH3 CH2CH3
-
What is the common name for dioxygen oxide?
-
11. Assume that the total cost function (TC) is given by the following equation: TC = 100+ 2.5y + 0.05 y2, where y is output. a. What is the total fixed cost (TFC)? b. What is the average total cost...
-
For the data set 8 6 14 4 (a) Calculate the deviations (x - ) and check to see that they add up to 0. (b) Calculate the variance and the standard deviation. 8 6 14 4
-
Consider an open loop system having a transfer function \(G(s)=\) \((s+1)^{-1}\). Find its output \(c(t)\) in BPF domain for a step input \(u(t)\) using the convolution matrix. Consider \(m=4\) and...
-
Plaintiffs operated a concession foods business called Festival Foods that provided food services at carnivals and festivals. The tangible assets of the business included a truck and servicing...
-
The cheque-clearing office of Pay Loans Company is responsible for processing all cheques that come to the company for payment. Managers at the company believe that variable over- head costs are...
-
1. 2. 3. (c) 11 Let f : R R be a continuous function such that f(3x)-f(x) =. If f(8) = 7, then f(14) is equal to: (a) 4 (b) 10 (d) 16 Let O be the origin and A be the point z = 1 + 2i. If B is the...
-
Balance in the General ledger of Andrea and Anne at the financial year end 30 September 2022 Account Capital Andrea (1/10/2021) Capital Anne (1/10/2021) Current Account Andrea(1/10/2021) Current...
-
What is the common name of each of the following compounds? a. CH 3 CH 2 OCH 2 CH 2 CH 3 b. CH3 -; CH3
-
Identify each of the following compounds as a primary, secondary, or tertiary amine, or as an amide. a. b. CH 3 CH 2 NHCH 2 CH 3 CH;NH
-
What is a semi-fixed cost?
-
How can we navigate the ethical complexities of globalized supply chains, including issues of labor rights, environmental impact, and economic inequality, in the context of increasingly...
-
What is the relationship among Expected rate of return, Expected dividend yield and the Expected growth rate (or the capital gains yield)? Explain in a a paragraph.
-
Construct the current assets section of the balance sheet from the following data. ( Use 3 6 5 days in a year. Do not round intermediate calculations. Round the final answers to the nearest whole...
-
An apparel manufacturer sells to a retail outlet on credit terms of net 45. The manufacturer's experience suggests payment is received and applied to the retailer's account 50 days after the invoice...
-
An aerobatic airplane has a key structural part of the empennage designed to undergo a design limit load of 7550 N at the operational maximum g- capability of the aircraft of 4.2 g. This part is made...
-
A hydrogen atom is in the ground state. It absorbs energy and makes a transition to the n = 3 excited state. The atom returns to the ground state by emitting two photons. What are their wavelengths?
-
Refer to the table to answer the following questions. Year Nominal GDP (in billions) Total Federal Spending (in billions) Real GDP (in billions) Real Federal Spending (in billions) 2000 9,817 578...
-
A rigid tank A of volume 0.6 m3 contains 3 kg water at 120oC and the rigid tank B is 0.4 m3 with water at 600 kPa, 200oC. They are connected to a piston cylinder initially empty with closed valves....
-
A cylinder containing 1 kg of ammonia has an externally loaded piston. Initially the ammonia is at 2 MPa, 180C and is now cooled to saturated vapor at 40C, and then further cooled to 20C, at which...
-
Calculate the heat transfer for the process described in Problem 4.70. A piston cylinder setup similar to Problem 4.24 contains 0.1 kg saturated liquid and vapor water at 100 kPa with quality 25%....
-
Describe Japan in terms of abundance and scarcity of factors of production in broad terms as well as in terms of, for example, types of skills of the labor force. Relate the description of these...
-
Draw a correctly labeled short-run aggregate demand and supply graph showing the equilibrium real GDP and price level, labeled Y, and P1. a. Show the effect of depreciation of the domestic currency..
-
Why has the Treasury and the FED sold bonds in the past if not to borrow? Why does Kelton say that this is no longer relevant?

Study smarter with the SolutionInn App



