When the following compound is treated with concentrated HCl at 100ºC for several hours, hydrolysis occurs, producing
Question:
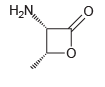
Transcribed Image Text:
H,N,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
HN HCI H...View the full answer

Answered By

Vikash Gupta
I am graduated in Physics in 2018, from KIRORIMAL COLLEGE, University of Delhi. Now I am persuing Master's degree in physics. I like to do physics problems. I have experience of 1 year in tutoring. I think Physics is the only subject where you understand things,how they are happening . In physics you learn Maths and apply it. So I would like to join your platform to solve many Physics problems.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What product is formed when the following compound is treated with Ag2O? HO
-
When the following compound is treated with sodium methoxide in methanol, two elimination products are possible. Explain why the deuterated product predominates by about a 7:1 ratio (refer to Problem...
-
Only a substitution product is obtained when the following compound is treated with sodium methoxide: Explain why an elimination product is not obtained. CH3 Br CH3
-
6. Kindly describe the accounting framework. What are the elements and how do they effect the financial statements? 7. What does the accounting model specify? What are the elements involved?
-
The Finishing Department of Lee and Lewis, Inc., the last department in the manufacturing process, incurred production costs of $220,000 during the month of June. If the June 1 balance in...
-
Dexall Company recently had a fire in its store. Management must determine the inventory loss for the insurance company. Since the firm did not have perpetual inventory records, the insurance company...
-
A \(2000-\mathrm{kg}\) car burns gasoline with \(25 \%\) efficiency. Accelerating from rest, how fast would this car go upon burning \(0.040 \mathrm{~L}\) of gasoline if \(1.0 \mathrm{~L}\) of...
-
This problem continues the Draper Consulting, Inc., situation from Problem 6-45 of Chapter 6. Draper performs systems consulting. Draper's February Cash from its general ledger is as follows:...
-
Assume a hypothetical age that has not been assumed by other students. Assume you would like to invest in a zero-coupon bond in order to have $1000,000 in your retirement account when you get to the...
-
Jennifer is the sales manager for AT&T for the state of Missouri. There are major sales offices in Columbia, Kansas City, St. Joseph, St. Louis, and Springfield. The distances in miles between these...
-
We saw in Section 25.6 that DCC can be used to form a peptide bond. We explored the mechanism, and we saw that DCC activates the COOH moiety so that it readily undergoes nucleophilic acyl...
-
Waxes can be hydrolyzed to yield an alcohol and a carboxylic acid. Draw the products obtained when triacontyl hexadecanoate undergoes hydrolysis.
-
Evaluate the evolution of Walmart's marketing campaign and tagline over the years. What does the company continue to do well? What are the pros and cons of its most recent strategic marketing plan?
-
examine managerial economic principles and standards that come into play on the issue of The impact of the COVID-19 pandemic on consumer demand for home exercise equipment ?
-
Unit 1.2 DB: Welfare Economy of the U.S. COLLAPSE How have social welfare programs in the US changed over time? How have changes in government policy, private sector involvement, and social attitudes...
-
a. Discuss the relationship between GDP, GDP per capita and population growth rate. Start with your hypothesis about what you expect this relationship to be, and why. Include and describe a ten-year...
-
7. Solve the linear equation 3x = 24 +7x for x.
-
An ARM may also be referred to as a floating payment loan. True or false
-
Find the slopeintercept form of the equation of the line containing the points (-2, 4) and (6, 8).
-
Which of the following is FALSE regarding the purchasing power parity (PPP). a. The PPP is a manifestation of the law of one price b. The PPP says that a country with a higher expected inflation can...
-
Addition of 1-bromobut-2-ene to magnesium metal in dry ether results in formation of a Grignard reagent. Addition of water to this Grignard reagent gives a mixture of but-1-ene and but-2-ene (cis and...
-
Show how you might synthesize the following compounds starting with alkyl, alkenyl, or aryl halides containing four carbon atoms or fewer. (a) 3-phenylprop-1-ene (b) 5-methylhex-2-ene c) dec-5-ene
-
Predict the products of the following proposed Diels-Alder reactions. (a) (b) (c) (d) (e) (f) CHO C-C-IC-C CN NC CN O + OCHs CN CH,O CN
-
Explain "who" pays the cost of government regulation of business. Be very specific in your answer. Distinguish between compliance costs and non-compliance costs. What happens when the costs cannot be...
-
Vanessa, aged 59, is retired and has a superannuation balance of $550,000. This balance includes a tax-free component of $175,000. If Vanessa makes a lump sum withdrawal of $175,000, what are the tax...
-
Assume Thich Nguyen's Meditation LLP had SUTA wages equal to FUTA wages in the current year of $415,669. Their SUTA rate = 4.3%. Gross wages = $900,500. What is their total unemployment taxes for the...

Study smarter with the SolutionInn App


