Which of the following amino acids has a polar side chain? NH2 HANCH-CH-CH,CH-CH NH2 CHHH CH CH3
Question:
Which of the following amino acids has a polar side chain?
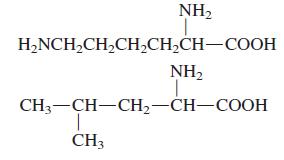
Transcribed Image Text:
NH2 HANCH-CH-CH,CH-CH—СООН NH2 CH—СH—СH —CH—СООН CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
Amino acids may have polar or non polar side chainS...View the full answer

Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following amino acids has a nonpolar side chain? NH2 CHSCH2CH2CH COOH NH2 HSCH CH COOH
-
Which of the following amino acids are likely to be linked to only one species of tRNA? What are their anticodons? (a) Phe (b) Leu (c) His
-
Show how you could prepare the following -amino acids from the appropriate carboxylic acids: (a) Phenylalanine (b) Valine
-
1. As a policy maker you should never worry much about those are eligible for Medicaid benefits and do not enroll. This is because they will enroll in public insurance if they need it. True or False?...
-
With reference to the extracurricular activities data in Exercise 2.3, obtain the (a) Sample mean. (b) Sample median. (c) Comment on the effect of a large observation.
-
A company that manufactures classroom chairs for high school students claims that the mean breaking strength of the chairs is 300 pounds. One of the chairs collapsed beneath a 220-pound student last...
-
Table B. 11 presents 38 observations on wine quality. a. Select four observations at random from this data set, then delete these observations and fit a model involving only the regressor flavor and...
-
Goliath Corporation issues $300,000 of bonds for $312,000. (a) Prepare the journal entry to record the issuance of the bonds, and (b) Show how the bonds would be reported on the balance sheet at the...
-
Can you determine whether the below actions ( i ) to ( v ) can be categorized as a ) Tax Planning, b ) Tax Management, orc ) Tax Evasion, providing reasons for eachi. Mr . Sarthak deposits 1 , 2 5 ,...
-
At December 31, 2022, Bramble Corporation reported the following plant assets. Land $ 4,278,000 Buildings $26,530,000 Less: Accumulated depreciation? buildings 17,005,050 9,524,950 Equipment...
-
If a sample of DNA isolated from a microorganism culture were analyzed and found to contain 1.5 mol of cytosine nucleotides and 0.5 mol of adenosine nucleotides, what would be the amounts of guanine...
-
Draw the zwitterion structure for the amino acid serine.
-
Duck, an accrual basis corporation, sponsored a rock concert on December 29, 2015. Gross receipts were $300,000. The following expenses were incurred and paid as indicated: Because the coliseum was...
-
Implement a static generic method that, given a Map , yields a List of the key/value pairs in the map.
-
Write a generic static method print that prints the elements of any object that implements the Iterable interface. The elements should be separated by commas. Place your method into an appropriate...
-
Modify Exercise P16.14 to use double hashing. The ith index in the probing sequence is computed as (h + i h2(k)) % L, where k is the original hash key before compression and h2 is a function mapping...
-
Read all words from a file and add them to a map whose keys are the first letters of the words and whose values are sets of words that start with that same letter. Then print out the word sets in...
-
Implement the following sorting algorithm. First split the given array a into nondecreasing and decreasing segments (that is, segments such that a[i] a[i + 1] ... a[i + k] or a[j] > a[j + 1] > ......
-
Norma, who uses the cash method of accounting, lives in a state that imposes an income tax. In April 2015, she files her state income tax return for 2014 and pays an additional $1,000 in state income...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
A spring loaded piston/cylinder contains 1.5 kg of air at 27C and 160 kPa. It is now heated to 900 K in a process where the pressure is linear in volume to a final volume of twice the initial volume....
-
Air in a piston/cylinder at 200 kPa, 600 K, is expanded in a constant-pressure process to twice the initial volume (state 2), shown in Fig. P5.101. The piston is then locked with a pin and heat is...
-
A vertical piston/cylinder has a linear spring mounted as shown so at zero cylinder volume a balancing pressure inside is zero. The cylinder contains 0.25 kg air at 500 kPa, 27oC. Heat is now added...
-
Explain possible advantages and disadvantages of PSL's current international trading strategy (subsidiary in China, export & import and online presence).
-
In November 2020 you entered into four May 2021 long futures contracts for crude oil (1,000 barrels per contract) for $65.00 per barrel. If the futures price was $70.00 per barrel on December 31,...
-
4) Champagne, Inc., had revenues of $13 million, cash operating expenses of $9 million, and depreciation and amortization of $2 million during 2008. The firm purchased $650,000 of equipment during...

Study smarter with the SolutionInn App


