A likely mechanism for the photolysis of acetaldehyde is Derive the rate law expression for the formation
Question:
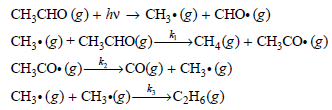
Derive the rate law expression for the formation of CO(g) based on this mechanism.
Transcribed Image Text:
CH CHO (s) + iv CH;-(g) + CНО-(g) CH,- (9) + сн,сноe) —А усH,(8) + сH,Cо- (g) →CH¼(g)+ CH;CO• CH;CO. (g)- CO(g) + CH3• (g) CH- (8) + сHз-(8)-3 сH(8)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
The rate is dCOdt k 2 CH 3 CO Applying the steadystate approximat...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Outline a likely mechanism for the solvomercuration step of the ether synthesis just shown. (b) Show how you would use solvomercuration-demercuration to prepare tertbutyl methyl ether. (c) Why...
-
Basing your answer on reactions that you have seen before, propose a likely mechanism for the condensation reaction in the first step of the preceding uridine synthesis.
-
Melphalan is a chemotherapy drug used in the treatment of multiple myeloma and ovarian cancer. Melphalan is an alkylating agent belonging to the nitrogen mustard family. Draw a likely mechanism for...
-
Which statement about the pass-by-reference is NOT true? a.Every time you pass a reference variable to a method, you also pass the object referred by the refenerece variable to the method. b.In the...
-
The cost C for a business to make personalized T-shirts is given by C(x) = 7.50x + 1500 where x represents the number of T-shirts. (a) The graphs of C and C1 are shown below. Match each function with...
-
(LO3) Which form is used to file a voluntary request to change an accounting method?
-
Estimate the parameters in a model for the gasoline mileage data in Table B. 3 using ridge regression with the value of \(k\) determined by Eq. (9.8). Does this model differ dramatically from the one...
-
Using property she inherited, Myrna makes a gift of $6.2 million to her adult daughter, Doris. The gift takes place in 2015. Neither Myrna nor her husband, Greg, have made any prior taxable gifts....
-
Solve the following LP using the simplex method. 4x1 + 2x2 maximize subject to -x1 + x2 < 2 4x1 x24 x1, x2 0 Use the Danzig rule (pick a variable with the most negative coefficient in z-row) to...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
Sunburn is caused primarily by sunlight in what is known as the UVB band, or the wavelength range from 290 to 320 nm. The minimum dose of radiation needed to create a sunburn (erythema) is known as...
-
If f = 1 10 10 s and k ic = 5 10 8 s 1 , what is f ? Assume that the rate constants for intersystem crossing and quenching are sufficiently small that these processes can be neglected.
-
Another type of system that is employed in engineering work is a group of parallel components or a parallel system. In this more conservative approach, the probability that the system operates is...
-
If D1 5 $2.00, g 5 6%, and P0 5 $40.00, what are the stocks expected dividend yield, capital gains yield, and total expected return for the coming year? Whereas a bond contains a promise to pay...
-
Firm A is expected to pay a dividend of $1.00 at the end of the year. The required rate of return is r s = 11%. Other things held constant, what would the stocks price be if the growth rate was 5%?...
-
Describe how the formula for a zero growth stock can be derived from the formula for a normal constant growth stock.
-
Write out and explain the valuation formula for a constant growth stock.
-
What would happen to a stocks price if the marginal investor examined a stock and concluded that its intrinsic value was greater than its current market price? Whereas a bond contains a promise to...
-
Company A, a calendar year taxpayer, has always used the cash method of accounting. It completed an engagement for a major client in November 2018 and submitted a bill for its $160,000 fee. Because...
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
Propose a plausible mechanism for each of the following transformations. a. b. c. d. e. f. OH 1) EtMgBr 2) H20
-
What product do you expect when tetrahydrofuran is heated in the presence of excess HBr?
-
Compound B has molecular formula C 6 H 10 O and does not possess any bonds. When treated with concentrated HBr, cis-1, 4-dibromocyclohexane is produced. Identify the structure of compound B.
-
Using Regression to Calculate Fixed Cost, Calculate the Variable Rate, Construct a Cost Formula, and Determine Budgeted Cost Pizza Vesuvio makes specialty pizzas. Data for the past 8 months were...
-
Mackenzie Corp. is preparing the December 31, 2023, year-end financial statements. Following are selected unadjusted account balances: Estimated warranty liability $ 6,650 Income tax expense Mortgage...
-
Kubin Company's relevant range of production is 20,000 to 23,000 units. When it produces and sells 21,500 units, its average costs per unit are as follows: Average Cost per Unit Direct materials $...

Study smarter with the SolutionInn App


