A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use
Question:
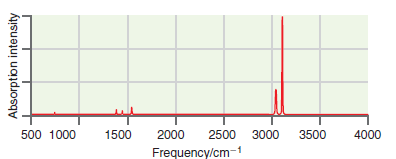
Transcribed Image Text:
500 1000 1500 2000 4000 2500 3000 3500 Frequency/cm-1 Absorption intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The peaks near 3100 cm 1 are indicative of CH stre...View the full answer

Answered By

Leah Muchiri
I am graduate in Bachelor of Actuarial Science and a certified accountant. I am also a prolific writer with six years experience in academic writing. My working principle are being timely and delivering 100% plagiarized free work. I usually present a precised solution to every work am assigned to do. Most of my student earn A++ GRADE using my precised and correct solutions.
4.90+
52+ Reviews
125+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A simulated infrared absorption spectrum of a gas phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
The investment shown in the following figure has an annual equivalent worth of $200 at i = 8%. Determine the cash flows in periods 2, 3, 5 and 6. $400 $400 3 4 5 6. Years $2,145
-
A tensile testing machine is shown in the following figure to determine the behavior of materials as they are deformed. (a) Use the provided data to calculate the stress and the corresponding strain...
-
A charge q is placed a distance from the origin, and a charge 2q is placed a distance 2r. There is a charge Q at the origin. If a charges are positive, which charge is at the higher potential? A q B)...
-
The equation of a standing wave is obtained by adding the displacements of two waves traveling in opposite directions (see figure). Assume that each of the waves has amplitude A, period T, and...
-
Andrea Pafko, a fellow student, contends that the double entry system means that each transaction must be recorded twice. Is Andrea correct? Explain
-
Bolt Industries is facing increased competition and wants to borrow $10 million in cash to protect against future revenue shortfalls. Currently, long-term AA rates are 10%. Given its credit rating,...
-
The Lynn Company uses a normal job-costing system at its Minneapolis plant. The plant has a machining department and an assembly department. Its job-costing system has two direct-cost categories...
-
i. As at 30th June 2013, the following information was available from the records of SGR Limited. ii. Acheque of sh 2,720,000 drawn on 30th June was presented to the bank for payment on 16th July,...
-
Consider the following 0x86 program: .data array DWORD 1,2,3,4,5,6,7,8,9 aravSize -(S-array)/4 ; array .code main PROC mov ecx,arraySize-1 mov esi.OFFSET array L1: mov eax. [esi] sda mov bx,2 idiv bx...
-
Fill in the missing step in the derivation that led to the calculation of the spectral line shape in Figure 19.24. Starting from and neglecting the first term in the parentheses, show that Figure...
-
Isotopic substitution is used to identify characteristic groups in an unknown compound using vibrational spectroscopy. Consider the C~C bond in ethane ( 12 C 2 1 H 6 ). By what factor would the...
-
Describe one or two lessons from Lockheed Martins Skunk Works that can be applied to implementing marketing programs.
-
What are Saturn's rings does geyser activity on Enceladus contribute toward?
-
Complete the following methods with the given signatures. All user input and output must occur in your main method. For this program, you may use Scanner or JOptionPane. In your main method, display...
-
Which elements exhibit an ethereal, fantastical quality, reminiscent of the realm of dreams or the realm of imagination?
-
After-tax Salvage Value Year 5-year 1 23 20.00% 32.00% 19.20% 4 11.52% 5 11.52% 6 5.76% The Honey Bee Co. purchased some equipment three years ago at a cost of $36,500. The equipment is 5-year...
-
On March 1, 2021, Planet Co. acquired 80% of Star Corp's outstanding common shares for $3,600,000. On the date of acquisition, the book value of Star Corp's net assets was $3,850,000. Book value...
-
A large blower for a furnace delivers 47000 ft 3 /min (CFM) of air having a specific weight of 0.075 lb/ft 3 . Calculate the weight flow rate and mass flow rate.
-
Prairie Outfitters, Inc., a retailer, accepts paymnent through credit cards. During August, credit card sales amounted to $12,000. The processor charges a 3% fee. Assuming that the credit card...
-
Before a rotameter can be used to measure an unknown flow rate, a calibration curve of flow rate versus rotameter reading must be prepared. A calibration technique for liquids is illustrated below. A...
-
How many of the following are found in 15.0 kmol of xylene (C 8 H 10 )? (a) kg C 8 H 1 ; (b) mol C 8 H 1 ; (c) lb-mole C 8 H 1 ; (d) mol (g-atom) C; (e) mol H; (f) g C; (g) g H; (h) molecules of C 8...
-
Certain solid substances, known as hydrated compounds, have well-defined molecular ratios of water to some other species. For example, calcium sulfate dihydrate (commonly known as gypsum, (CaSO 4 2H...
-
Briefly define TinyOS and explain its goals as an embedded operating system.?
-
Consider the graph shown on the right. Find the strongly connected components of the graph. Whenever you use DFS and there is a choice of nodes to explore, always pick the one that is alphabetically...
-
After viewing the previous video, "Types of Operating Systems As Fast As Possible", explain the difference between embedded operating systems and desktop operating systems. (300-yep, you guessed it)...

Study smarter with the SolutionInn App


