A stream of carbon monoxide flowing at 300 kg/min is cooled from 450C to 50C at a
Question:
A stream of carbon monoxide flowing at 300 kg/min is cooled from 450°C to 50°C at a low pressure.
(a) Calculate the required rate of cooling (kW), using the enthalpy function of APEx. Check the calculation using the heat capacity formula in Table B.2.
(b) State all the assumptions you made in Part (a). Where in the calculation did you make use of the fact that the cooling was done at a low pressure?
Table B.2
![TABLE B.2 Heat Capacities Form 1: C,[kJ/(mol-C)] or [kJ/(mol·K)] = a + bT + cT + dT Form 2: C,[kJ/(mol-C)] or [kJ/(mol-K)] = a + bT + cT? Example: (C,)acetone(e) = 0.07196 + (20.10 x 10-$)r - (12.78 x 10-8)7? + (34.76 x 10-12)Tr*, where T is in °C. %3D](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1590/0/5/6/4075ec655d7a186e1590056399297.jpg)
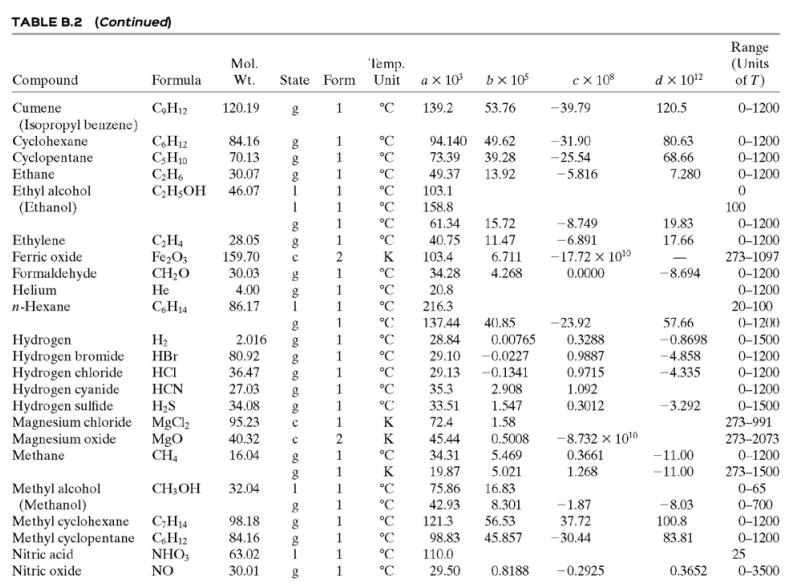
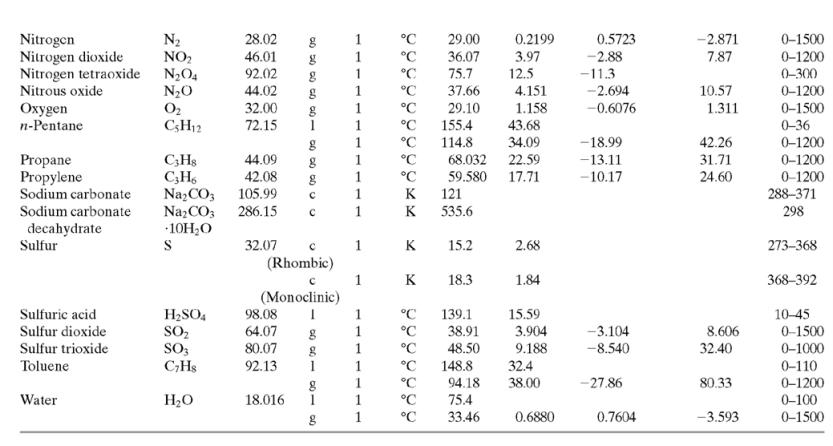
TABLE B.2 Heat Capacities Form 1: C,[kJ/(mol-C)] or [kJ/(mol·K)] = a + bT + cT + dT Form 2: C,[kJ/(mol-C)] or [kJ/(mol-K)] = a + bT + cT? Example: (C,)acetone(e) = 0.07196 + (20.10 x 10-$)r - (12.78 x 10-8)7? + (34.76 x 10-12)Tr*, where T is in °C. %3D Note: The formulas for gases are strictly applicable at pressures low enough for the ideal-gas equation of state to apply. Range (Units of T) Mol. Temp. State Form Unit ax 10 b x 10 ex 10 d x 1012 Compound Formula Wt. CH;COCH; 58.08 1 °C 123.0 -30-60 18.6 20.10 6.053 0.4147 Acetone °C 34.76 71.96 42.43 28.94 -12.78 -5.033 0.3191 0-1200 0-1200 0-1500 C;H2 26.04 29.0 °C Acetylene Air 18.20 -1.965 1.965 1. 28.09 0.1965 0.4799 273-1800 Ammonia NH, 17.03 °C 35.15 2.954 0.4421 -6.686 0-1200 Ammonium sulfate Benzene (NH4)2SO4 132.15 C,H, 1 215.9 275-328 126.5 74.06 78.11 1 °C 23.4 32.95 6-67 0-1200 1 -25.20 77.57 CH10 CH10 CHs CaC2 CACO, Ca(OH)2 Isobutane 58.12 89.46 49.87 30.13 27.88 -18.91 0-1200 0-1200 0-1200 n-Butane 58.12 92.30 -15.47 34.98 25.64 50.50 Isobutene Calcium carbide Calcium carbonate 56.10 1 82.88 -17.27 -8.66 x 1010 -12.87 x 1010 64.10 K 68.62 1.19 298-720 100.09 K 82.34 4.975 273-1033 Calcium hydroxide 74.10 K. 89.5 276-373 с Calcium oxide Carbon -4.52 x 1010 -4.891 x 100 -2.887 Cao 56.08 с K 41.84 2.03 273-1173 12.01 11.18 1.095 273-1373 с CO2 CO Carbon dioxide 44.01 1. 1. 1 36.11 4.233 7.464 0-1500 Carbon monoxide 28.01 28.95 0.4110 0.3548 -2.220 0-1500 Carbon tetrachloride CC4 Chlorine 153.84 70.91 63.54 93.39 12.98 273-343 Ch 1 °C 33.60 1.367 -1.607 6.473 0-1200 Соpper Cu 1. K 22.76 0.6117 273-1357
Step by Step Answer:
Ans A Q 145965874762913kJsec Explanation Given Mass flow of COCarbon monoxide 300kgmin or 300 103 gm...View the full answer



Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
A stream of nitrogen flowing at the rate of 2 kg s-1 and a stream of hydrogen flowing at the rate of 0.5 kg s-l mix adiabatically in a steady-flow process. If the gases are assumed ideal, what is the...
-
A saturated stream of carbon dioxide enters a dehumidifier with a flow rate of 100 m3/min at 39oC, 100 kPa. The mixture is cooled to 10oC by circulating cold water before being electrically heated...
-
A mixture of ideal gases flowing at 50 kg/min is compressed from 100 kPa and 20 8 C to 600 kPa in an insulated compressor. Determine the minimum horsepower requirement if the gravimetric analysis of...
-
In Fig. P2.72 gate AB is circular. Find the moment of the hydrostatic force on this gate about axis A. Neglect atmospheric pressure.
-
Draw the gel electrophoretic pattern that would be seen in dideoxy sequence analysis oî the DNA molecule in Problem 10. In Problem 10 5' GCAATCGTACGATTAGGGC 3' CGTTAGCATGCTAATCCCG
-
Retailers like RH create shopping themes that resemble a commercial living room where people can go to hang out and relax. This type of strategy converts the physical store into a __________. a....
-
For the purpose of calibration of measuring instrument, which of the following is used: (a) Screw gauge (b) Slip gauge (c) Dial gauge (d) Ion gauge
-
You will modify the Rounded Item Prices query to round retail values for items in the inventory. Open the Rounded Item Prices query in Design view. Create a new column to round the Retail price of...
-
Study Problem 9-8 (algo) Table A below shows abbreviated balance sheets for the central bank in the country of Beckland and B shows tables for its whole" commercial banking system. The target reserve...
-
(Multiple choice) (1) True or false: If the net torque on a rotating system is zero, the angular velocity of the system cannot change. (2) If the angular momentum of a system is constant, which of...
-
Use the enthalpy function of APEx to calculate H for each of the processes given below and check your solution to Part (a) by using the heat capacity tabulated in Table B.2. (Include units in your...
-
Among the best-known building blocks in nanotechnology applications are nanoparticles of noble metals. For example, colloidal suspensions of silver or gold nanoparticles (10200 nm) exhibit vivid...
-
The young minister was startled to learn that his parishioners considered him _______; he had been unaware that his message was being undermined by his sanctimonious and self-righteous tone. a....
-
The price system is based on _______. a) government regulation (i.e., the government sets most prices) b) the individual whim of the businessperson who sets it c) the feelings of the individual buyer...
-
Which statement is true? a) In recent years the U.S. economy has become primarily socialistic. b) There is one model for capitalism that virtually every capitalist economy follows closely. c) The...
-
Which of the following would be the best public policy? a) Zero tolerance for pollution. b) Allow private business firms to curb their own pollution. c) Provide business firms with incentives to curb...
-
Which statement is the most accurate? a) The South had some very substantial economic grievances against the North in the years immediately preceding the Civil War. b) The South seceded from the...
-
Which statement is true? a) Government failure, by definition, can occur only at the federal level. b) Nearly all the first responders to Hurricane Katrina in New Orleans were in the private sector....
-
In a Young's double-slit experiment that uses electrons, the angle that locates the first-order bright fringes is A = 1.6 10-4 degrees when the magnitude of the electron momentum is pA = 1.2 ...
-
Revol Industries manufactures plastic bottles for the food industry. On average, Revol pays $76 per ton for its plastics. Revol's waste-disposal company has increased its waste-disposal charge to $57...
-
Mandelate esters exhibit spasmolytic activity (they act as muscle relaxants). The nature of the alkyl group (R) greatly affects potency. Research indicates that the optimal potency is achieved when R...
-
Draw the alkoxide formed in each of the following cases: a. b. c. d. HO ? Na NaH
-
Identify whether each of the following compounds is chiral or achiral: a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. CI
-
Fill out the Special event template below. Directions : Conduct event-specific research to address the relevant areas of the outline. If some sections of the outline are not used, provide a brief...
-
A home-based sign company uses this function to model its monthly profit, where x is the price of each sign it sells. p(x)=-10x^(2)+498x-1,500 What is the company's profit if it sells each sign for...
-
Based on your analysis of the market data and a comprehensive understanding of the customer and buying processes, write a 2-3 page description of the major segments of the market and the specific...

Study smarter with the SolutionInn App


