Describe the changes you would observe as the temperature of a mixture of phenol and water at
Question:
Figure 9.21
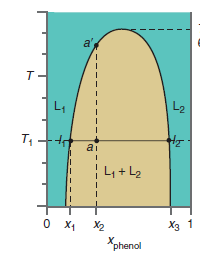
Transcribed Image Text:
a' L1 L2 ai L4 + L2 о х Хз 1 X2 Xphenol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
As T increases the number of moles in separate phases decreases un...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A red ball is placed at point A in FIGURE P34.44. a. How many images are seen by an observer at point O?b. What are the (x, y) coordinates of each image? 3.0 m 2.0 m 3.0 m 1.0 m FIGURE P34.44
-
What would be the final temperature of a mixture of 50 g of 20C water and 50 g of 40C water?
-
(a) Find the electric potential at point P in FIGURE 20-33. (b) Suppose the three charges shown in Figure 20-33 are held in place. A fourth charge, with a charge of +4.82 C and a mass of 2.33 g, is...
-
1. Enis falsely accuses Monalisa of stealing from Island Tours, Inc., their employer. Enis's statement is defamatory only if a. a third party hears it. b. Monalisa has not been caught. c. the...
-
A ratio is one value expressed to another. A financial ratio is one financial value or measurement expressed to another. There are about 20 financial ratios commonly used to assess one company's...
-
Consider the procedure for measuring a woman's center of gravity given in Example 8.3. The \(600 \mathrm{~N}\) woman is in place on the board, with the scale reading \(250 \mathrm{~N}\). She now...
-
The CEO of the company asks you to justify the 10 percent increase in sales training expenditures for next year. How would you satisfy this request by the CEO?
-
Oakwood Company produces maple bookcases to customer order. It received an order from a customer to produce 5,000 bookcases. The following information is available for the production of the...
-
Helen holds 2,400 shares of Fizbo Incorporated stock that she purchased 11 months ago. The stock has done very well and has appreciated $22/share since Helen bought the stock. When sold, the stock...
-
Locate all the instantaneous centres for the crossed four bar mechanism as shown in Fig. 6.28. The dimensions of various links are : CD = 65 mm; CA = 60 mm ; DB = 80 mm ; and AB = 55 mm. Find the...
-
An ideal dilute solution is formed by dissolving the solute A in the solvent B. Write expressions equivalent to Equations (9.9) through (9.13) for this case.
-
Describe the changes you would observe as the temperature of a mixture of triethylamine\ and water at point a in Figure 9.22 is increased until the system is at point a³. How does the relative...
-
An automobile tire manufacturer collected the data in the table relating tire pressure x (in pounds per square inch) and mileage (in thousands of miles): A mathematical model for the data is given by...
-
Implement SplitLists described in Exercise 3 as a client function. An Unsorted Type ADT is to be extended by the addition of function SplitLists, which has the following specifications: Exercise 3 1....
-
To tell if one element in ZooGraph has relation X to another element, you look for a path between them. Show whether the following statements are true, using the picture or adjacency matrix. 1....
-
Explain why software might need to be modified 1. in the design phase. 2. in the coding phase. 3. in the testing phase. 4. in the maintenance phase.
-
What C++ construct is used to represent ADTs?
-
Explain what we mean by software engineering.
-
Evaluate the following expression: 8 6 OrElse 4 < 7. a. True b. False
-
Illini Company, Inc. Balance Sheet as of 12/31/20X0 Assets Current Assets: Cash $1,500,000 Accounts receivable, net 18,000 Inventory 50,000 Total current assets 1,568,000 Equipment 90,000 Goodwill...
-
Balloons are still used to deploy sensors that monitor meteorological phenomena and the chemistry of the atmosphere. It is possible to investigate some of the technicalities of ballooning by using...
-
Derive an expression for the compression factor of a gas that obeys the equation of state p(V nb) = nRT, where b and R are constants. If the pressure and temperature are such that V m = 10b, what is...
-
A certain gas obeys the van der Waals equation with a = 0.50 m 6 Pa mol 2 . Its volume is found to be 5.00 10 4 m 3 mol 1 at 273 K and 3.0 MPa. From this information calculate the van der Waals...
-
Write an equation for the polynomial graphed below 5+ 4 + -5-4-3-2 y = 3. 2- 1 + + 2 3 4 5 -2 345 -5+
-
How can ethical leadership principles be leveraged to navigate the complexities of organizational dynamics and promote a culture of integrity, accountability, and social responsibility ?
-
Consider the 12-QAM constellation shown in Figure 2 and the baseband pulse, g(t) depicted in Figure 3. The 12 passband transmit waveforms are represented as Sm1(t) = Amig(t) cos (2fet) - Amag(t) sin...

Study smarter with the SolutionInn App


