Five cubic meters of a 1.00-molar aqueous sulfuric acid solution (SG = 1:064) is stored at 25C.
Question:
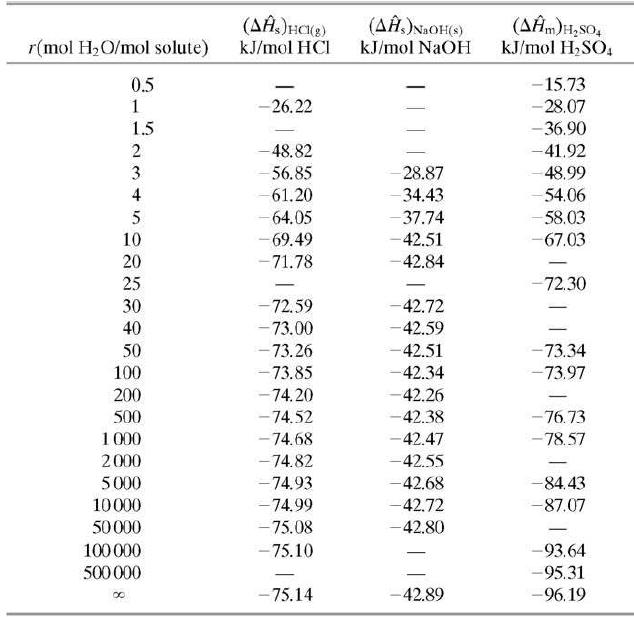
Five cubic meters of a 1.00-molar aqueous sulfuric acid solution (SG = 1:064) is stored at 25°C. Use data in Tables B.1 and B.11 to calculate the standard heat of formation of the solution in kJ/mol H2SO4 relative to the solute elements and water, and the total enthalpy of the solution relative to the same reference conditions.
Table B.11
Transcribed Image Text:
(AĤ.)NaOH(s) kJ/mol NaOH (AĤm)H,sO, kJ/mol H,SO, r(mol H2O/mol solute) kJ/mol HCI 0.5 -15.73 -26.22 -28.07 1.5 - 36.90 -48.82 -41.92 28.87 - 34.43 - 37.74 3 56.85 48.99 4 61.20 54.06 - 64.05 - 69.49 - 58.03 10 -42.51 -67.03 20 -71.78 42.84 - 25 -72.30 30 72.59 -42.72 - 73.00 - 73.26 - 73.85 -42.59 -42.51 -42.34 40 50 -73.34 100 -73.97 200 -74.20 -42.26 - 74.52 -74.68 - 76.73 - 78.57 500 -42.38 1 000 2000 -42.47 - 74.82 -42.55 5000 -74.93 -42.68 -84.43 10000 -74.99 -42.72 -87.07 50000 - 75.08 -42.80 100 000 - 75.10 -93.64 -95.31 -96.19 500 000 - -75.14 -42.89
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)

Answered By

Nicole omwa
Being a highly skilled tutor with at least 5 years of tutoring experience in different areas, I learned how to help diverse learners in writing drafts of research papers, actual research papers and locate credible sources. My assurance is built upon my varied knowledge of a variety of subjects. Furthermore, my involvement and interaction with numerous learners of all levels has allowed me to understand my clients' specific demands. Ultimately, this has aided me in being a better coach to learners to better their grades. Essentially, my responsibilities as a tutor would include:
Teaching abilities that assist pupils in enhancing their academic performance
Personal interaction with learners to make them understand abstract concepts
Inducing new skills and knowledge into their academic journeys
Fostering individual reflection, and independent and critical thinking
Editing and proofreading
Because I am constantly available to respond to your queries, you may decide to rely on me whenever you require my assistance. As an assurance, my knowledge skills and expertise enable me to quickly assist learners with different academic challenges in areas with difficulty in understanding. Ultimately, I believe that I am a reliable tutor concerned about my learner's needs and interests to solve their urgent projects. My purpose is always to assist them in comprehending abstract schoolwork and mastering their subjects. I also understand that plagiarism is a severe offense and has serious ramifications. Owing to this, I always make it a point to educate learners on the numerous strategies to have uniquely unique solutions. I am familiar with the following formatting styles:
MLA
APA
Harvard
Chicago
IEEE
Communication is always the key in every interaction with my learners. Hence, I provide timely communication about the progress of assigned projects. As a result, I make sure that I maintain excellent communication with all of my clients. I can engage with all of my customers more effectively, assisting them with their unique academic demands. Furthermore, I attempt to establish a solid working relationship with my leaners I have exceptional abilities in the below areas;
Sociology
History
Nursing
Psychology
Literature
Health and Medicine
Chemistry
Biology
Management
Marketing
Business
Earth Science
Environmental Studies
Education
Being a teacher who aces in diverse fields, I provide various academic tasks, which include;
Academic Reports
Movie Reviews
Literature Reviews
Annotated bibliographies
Lab reports
Discussion posts
Dissertations
Case study analyses
Research proposals
Argumentative Essays
I guarantee you high-quality Papers!!!!!
5.00+
17+ Reviews
32+ Question Solved
Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Question Posted:
Students also viewed these Sciences questions
-
Calculate the standard heat of formation of the compound ICl (g) at 25oC. Table s | 14 39 95 45 72 1 1419 6847064968 77386 42222 34 985 0302 121 Si H C O 437 490 9 31222241122 122221 25735 375859609...
-
A 10.0 mole% aqueous sulfuric acid solution (SG = 1.27) is to be titrated to neutrality at 25C with a 3.00-molar caustic soda (sodium hydroxide) solution (SG = 1.13): H 2 SO 4 (aq) + 2 NaOH (aq) Na 2...
-
A 5.00-wt% aqueous sulfuric acid solution (p = 1.03g/mL) flows through a 45-m long pipe with a 6.0cm diameter at a rate of 87L/min. (a) What is the morality of sulfuric acid in the solution? (b) How...
-
Let z(k) denote the k-year continuously compounded zero-coupon yield for the current term structure. You are given that z(1) = 0.035, z(2) = 0.041, z(3) = 0.045, z(4) = 0.049, z(5) = 0.051, z(6) =...
-
Is cognitive dissonance a good thing or a bad thing from an advertiser's point of view? Explain how and why advertisers should try to take advantage of the cognitive dissonance their consumers may be...
-
Show that the PSD function of a WSS random process \(\{X\}\) satisfies the following properties: (a) \(\Gamma_{X}(0)=\sum_{v=-\infty}^{\infty} R_{X}(v)\). (b) It is an even function; that is:...
-
Identify five defenses to an annulment action.
-
Refer to the information in Problem 20-51 for the Davidson Yachts Company. Required 1. What is the valuation of Davidson Yachts Company using the book value of equity method? 2. What is the valuation...
-
Take me to the text An employee had $21,800 in gross earnings up to March 20, 2021. She has the following information for her pay for the week ending March 27, 2021. Her employer contributes 100%...
-
Chicks with Kicks is a boxing club for women. The club manufactures all of its own punching bags out of high-quality leather and sand from Los Roques, one of Venezuela's most beautiful beaches. The...
-
Hydrogen is produced in the steam reforming of propane: The watergas shift reaction also takes place in the reactor, leading to the formation of additional hydrogen: The reaction is carried out over...
-
Five cubic meters of a 1.00-molar aqueous sulfuric acid solution (SG = 1:064) is stored at 25C. Use data in Tables B.1 and B.11 to calculate the standard heat of formation of the solution in kJ/mol H...
-
By what percentage could Bristol Myers earnings have declined in 2002 as a result of the fraud?
-
Is it true that negative correlation means that when one stock goes up the other goes down?
-
Do you agree that companies have a duty to stakeholders other than their shareholders? If so, justify who else they are obligated to and why.
-
Is it fair to utilize the least expensive capital resources, no matter where they come from? Why?
-
Portfolio efficiency Is a portfolio that provides the minimum standard deviation for any level of expected return efficient?
-
Is sustainability just a marketing tool to get people to think green, or is it a necessary business tool? Defend your answer.
-
A man holds a double-sided spherical mirror so that he is looking directly into its convex surface, 45 cm from his face. The magnification of the image of his face is +0.20. What will be the image...
-
Using thermodynamic data from Appendix 4, calculate G at 258C for the process: 2SO 2 (g) + O 2 (g) 88n 2SO 3 (g) where all gases are at 1.00 atm pressure. Also calculate DG8 at 258C for this same...
-
Provide at least five different acceptable IUPAC names for the following compound.
-
Compound A has molecular formula C 8 H 8 . When treated with excess Br 2 , compound A is converted into compound B, with molecular formula C 8 H 8 Br 2 . Identify the structures of compounds A and B....
-
In some circumstances, dehydrogenation is observed. Dehydrogenation involves the loss of two hydrogen atoms (the reverse of hydrogenation). Analyze each of the following dehydrogenation reactions and...
-
Q1) Find expressions for the Revenue, Cost, and Profit from selling x thousand items. Item Price Variable Cost $8.00 3816x Fixed Cost $71,174
-
1. There are many market disruptions in the news today. (If you get stuck, on the last slide of the PowerPoint, I've given you 2 examples.) Choose any disruptor in the news and, (a) summarize the...
-
Discuss at least five of the formatting decisions you would have to make when typing a table. Compare the decisions you provided to those of two of your classmates. Do you agree or disagree with the...

Study smarter with the SolutionInn App


