Identify the molecular orbitals for F 2 in the images shown here in terms of the two
Question:
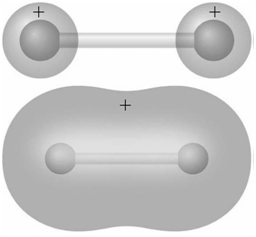
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The top image shows inphase AOs with no o...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Draw the molecular orbitals for the cyclopropenyl case. (Since there are three p orbitals, there must be three MOs: one all-bonding MO and one degenerate pair of MOs.) (b) Draw an energy diagram...
-
Evaluate the Klopp case in terms of those two criteria.
-
Images of molecular orbitals for LiH calculated using the minimal basis set are shown here. In these images, the smaller atom is H. The H1s AO has a lower energy than the Li2s AO. The energy of the...
-
If you could choose, which type of school would you want your imaginary child(ren) to attend?
-
The figure below shows the graph of y = sin (x - c) for c = - Ï/4, 0, and Ï/4. (a) How does the value of c affect the graph? (b) Which graph is equivalent to that of y = -cos (x +...
-
Portions of the payroll register for Barney's Bagels for the week ended July 15 are shown below. The SUTA tax rate is 5.4%, and the FUTA tax rate is 0.8%, both of which are levied on the first $7,000...
-
Carlos Martinez, who is a physical therapist, is thinking about starting a firm to provide in-home therapy services for people suffering from sports-related injuries. Carlos lives in Columbus, Ohio....
-
Sportz, Inc., manufactures athletic shoes and athletic clothing for both amateur and professional athletes. The company has two product lines (clothing and shoes), which are produced in separate...
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
Li Ming Yuan and Tiffany Shaden are the department heads for the accounting department and human resources department, respectively, at a large textile firm in the southern United States. They have...
-
Show that the Slater determinant formalism automatically incorporates the Pauli exclusion principle by evaluating the He ground-state wave function of Equation (21.9), giving both electrons the same...
-
Why are the magnitudes of the coefficients c a and c b in the H 2 + wave functions g and u equal?
-
The Walt Disney Company's annual report for the year ended October 3, 2015, includes income statements for the years ending in 2013, 2014, and 2015. Net income for these three years is as follows...
-
Account Cost of Goods Sold Net Sales Selling Expenses Amount (or percentage) $105,762,400 $314,756,900 $38,476,100 Interest Expense Administrative Expenses Common Stock Dividends Paid Income tax...
-
A manufacturer of self-sealing stem bolts finds that their product has a mean length of 15 cm and a standard deviation of 0.5cm. Suppose that the lengths follow a Normal distribution. What length...
-
you examine how budgets are developed. After developing a sample budget for a product or service, you also examine how ratios can be used to analyze the budget and provide information that can guide...
-
After accounting for the compressed volume loss loss on an adult patient being ventilated by a volume cycled vent, you still note a 200 ml difference between the expected and actual delivered volume....
-
What alternative data sources could be used in Step 1 of the problem-solving process if you only have access to office referrals?
-
An article in the New York Times in 2016 notes that both Hillary Clinton and Donald Trump want to label China as a currency manipulator. a. What is a currency manipulator? Why would China want to...
-
How can a promoter avoid personal liability for pre-incorporation contracts?
-
Calculate the pressure differential of water across the surface of a spherical droplet of radius 200 nm at 20C.
-
The contact angle for water on clean glass is close to zero. Calculate the surface tension of water at 20C given that at that temperature water climbs to a height of 4.96 cm in a clean glass...
-
Calculate the potential energy of the interaction between two linear quadrupoles when they are (a) Collinear, (b) Parallel and separated by a distance r.
-
If the local professional basketball team, the Sneakers, wins today's game, they have a 3 chance of winning their next game. If they lose this game, they have a chance of winning their next game. a)...
-
There is a function f of the form .12 f(x) = ax + x 13 for which f(0.1) = 6.06 10 and (0.9) = 0.03577. Determine a and B, and assess the sensitivity of these parameters to slight changes in the...
-
The order states: Give antihistamine elixir 2.5mg/kg/dose PO q4h. The patient weighs 143lbs. The drug is available as Antihistamine Elixir 200mg in every 8mL. Answer each of the following questions. ...

Study smarter with the SolutionInn App


