Show that the determinantal property used in the discussion of localized and delocalized orbitals in Section 24.6
Question:
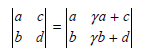
used in the discussion of localized and delocalized orbitals in Section 24.6 is correct.
Transcribed Image Text:
ya + c a ь la b yb + d 9.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
We first assume that for some con...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Why cant localized orbitals be represented in an MO energy diagram?
-
Why are localized and delocalized models equally valid for describing bonding in closed-shell molecules? Why cant experiments distinguish between these models?
-
The occupied MOs of ammonia are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
Terminology Key: Key Word List Describe Explain What to do In bulleted, numbered or paragraph form, provide a number of consecutive items-if paragraph form, use commas (,) to separate items In...
-
Find two solutions of each equation. Give your answers in degrees (0o < 360o) and in radians (0 < 2). Do not use a calculator. 1. (a) sin = 1/2 (b) sin = 1/2 2. (a) cos = 2/2 (b) cos = 2/2
-
WHETHER TO DISCONTINUE A DEPARTMENT Williams and Lloyd Company is trying to decide whether to discontinue department B. Operating results for the year just ended for each of the company's three...
-
Draw energy diagrams for the person and the package in Figure 10.21 . Figure 10.21 Two equivalent situations in which an object is accel- erated by static friction. (a) Person steps forward from...
-
Joakim Keynes is the manager of the Repairs and Maintenance Department of JB Industries. He is responsible for preparing his departments annual budget. Most managers in the company inflate their...
-
8.The following data is available: Country Y currency Dollar Country X currency Peso Country Y interest rate 1% per year Country X interest rate 3% per year Country X expected inflation rate 2% per...
-
A 100 kg block of solid is moved upward by an external force F as shown in the accompanying figure. After a displacement of 10 cm, the upper surface of the block reaches a linear spring at its rest...
-
A cyclic polyene is known to be non-planar. Are the MO energy levels of this molecule well described by the Huckel model? Justify your answer.
-
Predict whether LiH + 2 and NH 2 should be linear or bent based on the Walsh correlation diagram in Figure 24.11. Explain your answers. Figure 24.11 1b, + 2a, 1b2 1a1 tog 100 120 140 160 180 Bond...
-
Derive equation (25-11) for component A in terms of molar units, starting with the control-volume expression for the conservation of mass. CA - RA = 0 V NA +: (25-11)
-
Consider a project that requires an investment of $35,000 today and generates after-tax cash flows of $8,000 per year over the next 5 years. The appropriate discount rate is 13 percent. What is the...
-
A company's 2022 income statement showed depreciation expense of $900,000 and interest expense of $500,000. The addition to retained earnings for the year was $700,000 and dividends paid during the...
-
White has previously said that we need a new way to think about being old in America. She is calling on artists, change agents, and social entrepreneurs to help imagine a change that will better the...
-
BEAM047-Exam Paper_BEAM047R-AUG22 (1).pdf - Adobe Acrobat Reader DC (64-bit) File Edit View Sign Window Help Home Tools 26C Sunny BEAM047-Exam_Pa... x 2 / 4 125% wwww The possible returns of stocks...
-
James and Helena are 43 years old both and have three children. They have an annual household income of 85,000 and they have mortgage. With their current financial situation, what type of life...
-
What is pegging? What are the advantages of pegging? What are the disadvantages? Briefly discuss the controversy over Chinas management of the yuan-dollar exchange rate.
-
Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm (3 points) D=m/v...
-
What are the relative populations of the states of a two-level system when the temperature is infinite?
-
The element polonium crystallizes in a cubic system. Bragg reflections, with X-rays of wavelength 154 pm, occur at sin = 0.225, 0.316, and 0.388 from the (100), (110), and (111) sets of planes. The...
-
A 1.0 dm 3 glass bulb contains 1.0 10 2 3 H 2 molecules. If the pressure exerted by the gas is 100 kPa, what are (a) The temperature of the gas, (b) The root mean square speeds of the molecules? (c)...
-
1. Develop a definition for the Triple C model of project management. 2. List some of the factors that can impede the flow of information for project planning purposes. How can these factors be...
-
The Meat Mart has $900,000 in net income. The firm has 200,000 shares of stock outstanding. The market price per share is $76. What is the PE (price to earnings) ratio?
-
Cost of Goods Manufactured for a Manufacturing Company The following information is available for Fuller Manufacturing Company for the month ending January 31: Cost of direct materials used in...

Study smarter with the SolutionInn App


