The amino acid glycine dimerizes to form the dipeptide glycylglycine according to the reaction 2Glycine(s) Glycylglycine(s) +
Question:
2Glycine(s) †’ Glycylglycine(s) + H2O(l)
Calculate ΔS, ΔSsurroundings , and ΔSuniverse at T = 298 K. Useful thermodynamic data follow:
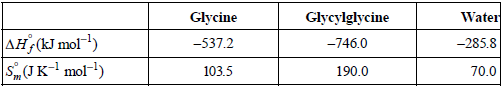
Transcribed Image Text:
Glycylglycine Water -285.8 Glycine -537.2 AH (kJ mol) SJK- mol-!) -746.0 103.5 190.0 70.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
ASR 2 1035JK mol 1900 J K mol700 JK mol 5...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The diffusion constant for the amino acid glycine in water has a value of 1.06 10-9 m2/s. In a 2.0-cm-long tube with a cross-sectional area of 1.5 10-4 m2, the mass rate of diffusion is m/t = 4.2 ...
-
The amino acid glycine can be condensed to form a polymer called polyglycine. Draw the repeating monomer unit.
-
The amino acid glycine (H2N-CH2-COOH) can participate in the following equilibria in water: (a) Use the values of Ka and Kb to estimate the equilibrium constant for the intramolecular proton transfer...
-
2. Consider two households, one with an income twice as large as the other. Both households pay the equivalent of 5% of their income to consume free (public) education to the quantity PS (where PS...
-
Your manager is quite impressed with the security considerations you explained to her thus far, however, she asks that you give further thought to additional security measures where possible and to...
-
Explain with examples each of the eight steps in the strategic management process.
-
Aaron loans Victoria \($10,000\) with interest compounded at a rate of 8% annually. How much will Victoria owe Aaron if she repays the entire loan at the end of five years?
-
Royale Aluminum desires an after-tax income of $500,000. It has fixed costs of $2,500,000, a unit sales price of $300, and unit variable costs of $150, and is in the 40% tax bracket. Required: 1....
-
Mickley Company's plantwide predetermined overhead rate is $18.00 per direct labor-hour and its direct labor wage rate is $11.00 per hour. The following information pertains to Job A-500: Direct...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Because V is a state function, ((V/T) P /P) T = ( (V/P) T /T) P . Using this relationship, show that the isothermal compressibility and isobaric expansion coefficient are related by ( /P) T = (/T) P .
-
How would you expect a park manager of a national park that adheres to natural regulation respond to a lightning-induced forest fire? To the establishment of a noxious invasive weed species?
-
Describe the internal and external forces for change.
-
What role does moral courage play in confronting ethical dilemmas and standing up for principles of justice and integrity, particularly in contexts where doing so entails personal sacrifice or facing...
-
How does the philosophical concept of existential courage inform our understanding of individuals' capacity to confront existential dilemmas and navigate the inherent uncertainties of human existence...
-
What impact did COVID have on the market for new cars? Why are we defining the market being studied as the market for used cars and not just cars?
-
Septer Corporation issued 2,000 of its $1,000, 8% ten-year bonds dated July 1,Year 1 on September 1, Year 1, at a time when the market paid 9% for bonds of similar risk. The bonds were quoted at 94...
-
In what ways do socio-cultural contexts shape the manifestation of courage, and how do cultural narratives and collective values influence individuals' willingness to take risks and challenge...
-
Determine the monthly payments of principal and inter-est for a 30-year loan. The Leungs decided to build a new house. The contractor quoted them a price of $144,500, including the lot. The taxes on...
-
Assume Eq. 6-14 gives the drag force on a pilot plus ejection seat just after they are ejected from a plane traveling horizontally at 1300 km/h. Assume also that the mass of the seat is equal to the...
-
Estimate the coefficients a and b in the Dieterici equation of state from the critical constants of xenon. Calculate the pressure exerted by 1.0 mol Xe when it is confined to 1.0 dm3 at 25C.
-
Given that Som = 29.79 J K-I mol-1 for bismuth at 100 K and the following tabulated heat capacities data (D.G. Archer,]. Chem. Eng. Data 40, 1015 (1995)), compute the standard molar entropy of...
-
Estimate the standard reaction Gibbs energy of N2 (g) + 3 H2 (g) t 2 NH3 (g) at (a) 500 K, (b) 1000 K from their values at 298 K.
-
PART A (Retained Earnings Statement) January 1, Retained Earnings Beginning Balance 2021 Income before income tax 1 $2,340,000 Prior period adjustment: understatement of 2020 depreciation expense...
-
There are 3 source and 4 destinations, For the data below, construct an initial feasible solution. Cost Destination 1 Destination 2 Destination 3 Destination 4 Supply Source 1 12 18 9 11 105 Source 2...
-
A company is using Dynamics 3 6 5 Project Operations. You acquire an independent software vendor ( ISV ) solution from AppSource that contains several project charts and install the solution in the...

Study smarter with the SolutionInn App


