The experimentally determined density of O 2 at 140. bar and 298 K is 192 g L
Question:
Figure 7.8
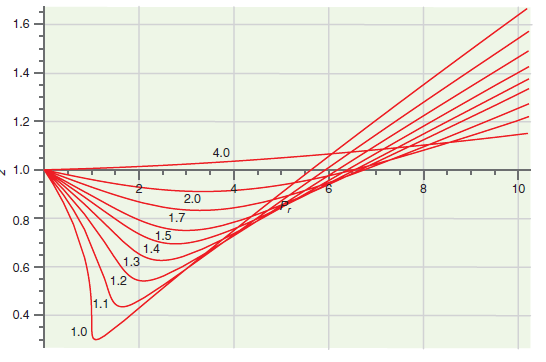
Transcribed Image Text:
1.6 1.4 1.2 4.0 v 1.0 8. 10 2.0 0.8 1.7 1.5 1.4 1.3 0.6 1.2 1.1 0.4 1.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Because P V 140 Bar5043 bar 278 and T V 298 ...View the full answer

Answered By

Rohail Amjad
Experienced Finance Guru have a full grip on various sectors, i.e Media, Insurance, Automobile, Rice and other Financial Services.
Have also served in Business Development Department as a Data Anlayst
4.70+
32+ Reviews
83+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At 725 K and 280. bar, the experimentally determined density of N 2 is 4.13 mol L 1 . Compare this with values calculated from the ideal and RedlichKwong equations of state. Use a numerical equation...
-
Calculate the density of O 2 (g) at 480. K and 280. bar using the ideal gas and the van der Waals equations of state. Use a numerical equation solver to solve the van der Waals equation for V m or...
-
For the 2-kg connecting rod shown, it has been experimentally determined that the mass moments of inertia of the rod with respect to the center-line axes of the bearings AA and BB are, respectively,...
-
Welch Company prepares monthly financial statements. Below are listed some selected accounts and their balances in the September 30 trial balance before any adjustments have been made for the month...
-
What are the advantages and disadvantages of the single-step income statement?
-
When a current is passed through the wire in Figure, will it tend to bunch up or form acircle? I
-
In the open wire loop of Figure P28.25, end 1 is held at a higher electric potential than end 2. (a) In which direction do electrons move through the loop? (b) What is the direction of the current?...
-
The Talley Corporation had a taxable income of $365,000 from operations after all operating costs but before (1) interest charges of $50,000, (2) dividends received of $15,000, (3) dividends paid of...
-
Fluorine Limited (FL), a manufacturer of ships, has entered into the following contracts during the year ended 31 December 2022: (i) On 1 January 2022, FL entered into a contract with Alpha Limited...
-
Suppose that the production function for DVDs is given by Q = KL2 L3, where Q is the number of disks produced per year, K is machine-hours of capital, and L is man-hours of labor. a) Suppose K =...
-
A business has assets of $1,000,000 and liabilities of $600,000. What is the amount of owners equity in this business? a. $400,000 b. $1,400,000 c. $600,000 d. $1,600,000
-
A 1.75 mole sample of Ar undergoes an isothermal reversible expansion from an initial volume of 2.00 L to a final volume of 85.00 L at 310. K. Calculate the work done in this process using the ideal...
-
Shant Peterson is an attorney in Los Angeles. Peterson Law Office uses the direct write-off method to account for uncollectible receivables. At April 30, 2025, Peterson Law Offices accounts...
-
Consider two variables, \(y\) and \(x\). Do a regression of \(y\) on \(x\) to get a slope coefficient that we call \(b_{1, x, y}\). Do another regression of \(x\) on \(y\) to get a slope coefficient...
-
A slab of clay \(50 \mathrm{~mm}\) thick was dried from both flat surfaces-with the four thin edges sealed-by exposure to dry air. The initial uniform moisture content was \(15 \%\). The drying took...
-
Aeration is a common industrial process and yet one in which there is often serious disagreement about correlations (Cussler, 1997). This is especially true for deep-bed fermentors and for sewage...
-
In a laboratory experiment, air at \(300 \mathrm{~K}\) and 1 atm is blown at high speed parallel to the surface of a rectangular shallow pan that contains liquid acetone \(\left(\mathrm{C}_{3}...
-
(a) Estimate the diffusivity of naphthalene \(\left(\mathrm{C}_{10} \mathrm{H}_{8} ight)\) in air at \(303 \mathrm{~K}\) and 1 bar. Compare it with the experimental value of \(0.087 \mathrm{~cm}^{2}...
-
Fill in the blanks with an appropriate word, phrase, or symbol(s). When the conclusion of an argument necessarily follows from the given set of premises it is a(n) __________ argument.
-
Pedro Bourbone is the founder and owner of a highly successful small business and, over the past several years, has accumulated a significant amount of personal wealth. His portfolio of stocks and...
-
Evaluate the triple integral in cylindrical polar coordinates: 3.00 4.00 I = zp cos? (4) d dp dz.
-
Find the volume of a right circular cylinder of radius a = 4.00 with a paraboloid of revolution scooped out of the top of it such that the top surface is given by z = 10.00 + 1.00 2 and the bottom...
-
Find the volume of a solid with vertical walls such that its base is a square in the xy plane defined by 0 x 2.00 and 0 x 2.00 and its top is defined by the plane z = 20.00 + x + y.
-
On January 1, 2019, Jerome Company purchased nontrading equity investments which are irrevocably designated at FVOCI Purchase Transaction price Market value December 31, 2019 cost Security A Security...
-
Identify the revision of the sentence that removes fillers without changing the meaning of the sentence. Isabella was looking for the gross income total in the report when she said she was absolutely...
-
You are considering investing in the following 10 year cash flow. If you required rate of return is 8%, what is the current value of this project? Assume all cash flows occur at the end of the year....

Study smarter with the SolutionInn App


