The Grotrian diagram in Figure 22.7 shows a number of allowed electronic transitions for He. Which of
Question:
a. 1s2 1S †’ 1s2p 1P
b. 1s2p 1P †’ 1s3s 1S
c. 1s2s 3S †’ 1s2p 3P
d. 1s2p 3P †’ 1s3d 3D
Figure 22.7
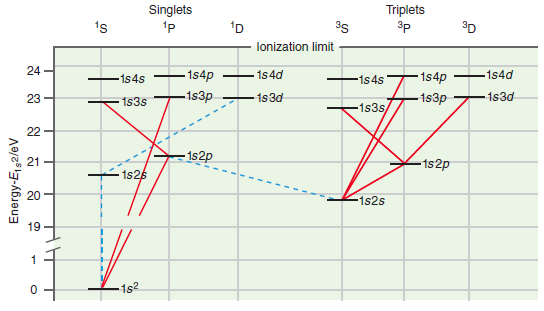
Transcribed Image Text:
Singlets Triplets 1s 35 3D lonization limit 24 - 1s4p 1s4d -1s4d - 1s4p -1s4s 1s4s -1s3p 1s3d 1s3d -1s3p 23 1s3s 1s3s, 22 - 152p 21 -1s2p 1s26 -1s2s 19 1s2 Energy-E92/eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
a 1s 2 1 S 1s2p 1 P No splitting because each term consists of a singl...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The figure below represents part of the emission spectrum for a one- electron ion in the gas phase. All the lines result from electronic transitions from excited states to the n = 3 state. a. What...
-
The following is an energy- level diagram illustrating three different electronic transitions in the Bohr hydrogen atom. a. Explain why the energy levels get closer together as they increase. Provide...
-
The energy-level diagram in Figure 9.36 shows that the sideways overlap of a pair of p orbitals produces two molecular orbitals, one bonding and one anti-bonding. In ethylene there is a pair of...
-
Reading material Raymond Frost, Alexa K. Fox & Judy Strauss (2019). Product: The Online Offer. E-Marketing , 9, 206 - 228. Raymond Frost, Alexa K. Fox & Judy Strauss (2019). Price: The Online Value....
-
Determine the angle between the diagonal of a cube and the diagonal of its base, as shown in the figure. a a a
-
Should the company proceed with plans to transition half of its employees to full-time remote work? Why or why not?
-
An instructor administers a 27 -item quiz to her class of 25 students. Each student's score on the quiz is the number of items answered correctly. These scores are listed below: a. Construct a...
-
Donald Fishing Charters has collected the following data for the December 31 adjusting entries: a. The company received its electric bill on December 20 for $ 400 but will not pay it until January 5....
-
Sage Inc. experienced the following transactions for Year 1, its first year of operations: 1. Issued common stock for $80,000 cash. 2. Purchased $180,000 of merchandise on account. 3. Sold...
-
Cheapo Electrons is an electricity retailer. The table below shows the load that it forecast its consumers would use over a 6-h period. Cheapo Electrons purchased in the forward market and the power...
-
In the Na absorption spectrum, the following transitions are observed: 4p 2 P 3s 2 S = 330.26 nm 3p 2 P 3s 2 S = 589.593 nm, 588.996 nm 5s 2 S 3p 2 P = 616.073 nm, 615.421 nm Calculate the...
-
List the quantum numbers L and S that are consistent with the following terms: a. 4 S b. 4 G c. 3 P d. 2 D
-
You are the manager of a local sporting goods store and recently purchased a shipment of 60 sets of skis and ski bindings at a total cost of $30,000 (your wholesale supplier would not let you...
-
Ecological interdependencies may be manifested in ways that are either competitive or symbiotic. However, what matters is the fact that interdependencies are essential for material cycling within a...
-
Compare PayPal and Google Wallet from a consumer perspective and from a merchant perspective.
-
What is an IPO?
-
Explain in your own words what exactly the concept of pollution-damage cost represents. Provide two examples.
-
An ecosystem is often described as the "house of life." This is in large part because al "living" ecosystems have to contain the six elements essential for life in some naturally prescribed manner....
-
In problem, solve each equation. log 6 36 = 5x + 3
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
Distinguish between semi-empirical, ab initio, and density functional theory methods of electronic structure determination.
-
What information does the term symbol 1 D 2 provide about the angular momentum of an atom?
-
Show by explicit integration that (a) Hydrogenic 1s and 2s orbitals, (b) 2p x and 2p y orbitals are mutually orthogonal.
-
Discuss the dynamic organization of the cytoskeleton and its pivotal role in cellular motility, intracellular transport, and structural integrity .
-
Would you support the idea of a government issued Digital currency? Why ? and why not?
-
To protect her savings against further inflation and to help her prepare for a healthy financial future, Hanna Lind deposits $9,100 in an investment account earning 6% interest compounded quarterly....

Study smarter with the SolutionInn App


