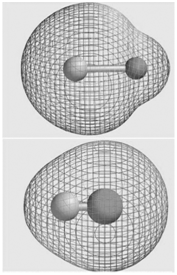
The molecular electrostatic potential maps for LiH and HF are shown here. Does the apparent size of
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Yes it does The electrostatic potential is displayed on a tot...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the molecular electrostatic potential map for the NH 3 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the BH 3 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the BeH 2 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
What was the action you are most proud of, or that is the most meaningful to you? What was the civic issue that was involved in your action? When and how did you first hear about that issue? Why did...
-
In Exercises 1-4, find the exact value of the expression, if possible. 1. sin(arctan 3/4) 2. cos(arcsin 4/5) 3. cos(tan 12) 4. sin(cos 15)
-
Elaborate three real-world examples of map, partition, and relation?
-
Consider the sample space \(S=\{y y, y n, n y, n n\}\) in Example 2.2. Suppose that the subset of outcomes for which at least one camera conforms is denoted as \(E_{1}\). Then, \[ E_{1}=\{y y, y n, n...
-
Harold, a CPA, has a new client who recently moved to town. Harold prepares the client's current-year tax return, which shows a net 1231 gain. Harold calls the client to request copies of the...
-
What are the influence tactics Managers choose ?
-
A seminar was recently attended by the Managing Director of XYZ Manufacturing Company Limited located at Sheffield. The focus of the seminar was "optimising scarce resources utility in a...
-
Why is it reasonable to approximate H 11 and H 22 by the appropriate ionization energy of the corresponding neutral atom?
-
For H + 2 , explain why H aa is the total energy of an undisturbed hydrogen atom separated from a bare proton by the distance R.
-
Mary Ott is going to borrow $10,400 for 120 days and pay $150 interest. What is the effective rate of interest if the loan is discounted?
-
Rawlsian justice is a philosophical argument in favor intergenerational fairness. What does intergenerational fairness mean in this context? How is this related to CBA? Explain.
-
What are the two items whose sum is the cost of equity?
-
How might the different ages of firms distort comparisons of their fixed assets turnover ratios?
-
Describe the differences between direct competitors, indirect competitors, and future competitors.
-
Discuss the most limiting assumptions on allocative efficiency in DEA.
-
Between 5:00 PM and 6:00 PM, cars arrive at McDonald's drive-thru at the rate of 20 cars per hour. The following formula from probability can be used to determine the probability that x cars will...
-
r = 0.18 Find the coefficients of determination and non-determination and explain the meaning of each.
-
The molar absorption coefficient of a substance dissolved in hexane is known to be 855 dm 3 mol 1 cm 1 at 270 nm. Calculate the percentage reduction in intensity when light of that wavelength passes...
-
The term symbol for the ground state of N 2 + is 2 g + . What is the total spin and total orbital angular momentum of the molecule? Show that the term symbol agrees with the electron configuration...
-
Explain the origin of the term symbol 3 g for the ground state of dioxygen.
-
calculate the payback period, internal rate of return, and net present value of the proposed mine
-
You need to choose between the following two design projects: year Cash flow A cash flow B 0 -$81,000 -$21,800 1 38,600 12,200 2 38,600 12,200 3 38,600 12,200 a) If the required return is 10 percent...
-
Home Furnish has bonds on the market with 13 years to maturity and a yield to maturity of 8.5.In the financial newspaper, Home Furnish bonds is quoted as 127.223% of par value. The bonds make...

Study smarter with the SolutionInn App


