The overall reaction for the halogenation of a hydrocarbon (RH) using Br as the halogen is RH
Question:
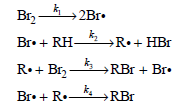
Determine the rate law predicted by this mechanism.
Transcribed Image Text:
Br2 2Br. Br• + RH- →R•+ HBr R•+ Brz -4 »RBr + Br• k. RBr Br• + R.-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
Applying the steadystate approximation to the intermediate species Br and ...View the full answer

Answered By

Anjali Arora
Having the experience of 16 years in providing the best solutions with a proven track record of technical contribution and appreciated for leadership in enhancing team productivity, deliverable quality, and customer satisfaction. Expertise in providing the solution in Computer Science, Management, Accounting, English, Statistics, and Maths.
Also, do website designing and Programming.
Having 7 yrs of Project Management experience.
100% satisfactory answers.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write the overall reaction for the fixation of nitrogen via the nitrogenase complex.
-
The overall reaction for the electrolytic production of aluminum by means of the Hall process may be represented as Al2O3 (s) + 3C (s) 2Al(l) + 3CO(g) At 1000C, the standard free-energy change for...
-
The overall reaction in the lead storage battery is Pb(s) + PbO2(s) + 2H+(aq) + 2HSO4-(aq) 2PbSO4(s) + 2H2O(l) d. Based on your previous answers, why does it seem that batteries fail more often on...
-
Below is the article related to the funding and construction of the SD Padres' ballpark in downtown San Diego. The article below relates to the Padres' decision to incur substantial payoff...
-
Restrict the domain of f (x) = x2 + 1 to x 0. Use a graphing utility to graph the function. Does the restricted function have an inverse function? Explain.
-
Using Table 1.3 of the IRS Statistics on Income Individual Income Tax Returns Publication (Complete Report 2020)...
-
Piecewise Linear Regression. In Example 7.3 we showed how a linear regression model with a change in slope at some point $t\left(x_{\min } Example 7.3 An important special case of practical interest...
-
The following table shows the transactions experienced by J G Gutter Works (JGGW) during Year 7. The table contains missing data which are labeled with alphabetic characters (a) through (j). Assume...
-
2. YA Y F FA 12 C 0=60 X In the figure, XY-coordinate system is the global coordinate System, and xy-coordinate system is the local coordinate system. The force vector at C on truss element is given...
-
Suppose a final exam contains 120 multiple-choice questions with 5 choices for each answer, and a poorly prepared student knows the answers to 60 questions and guesses on the other 60. What is the...
-
The enzyme fumarase catalyzes the hydrolysis of fumarate: Fumarate (aq) + H 2 O(l) L-malate (aq) The turnover number for this enzyme is 2.5 10 3 s 1 , and the Michaelis constant is 4.2 10 6 M....
-
Consider the collision-induced dissociation of N2O5(g) via the following mechanism: The asterisk in the first reaction indicates that the reactant is activated through collision. Experimentally it is...
-
Discuss why and how investors can use trend-following strategies as a hedge against inflation.
-
What are the relevant cash flows for an international investmentthe cash flows produced by the subsidiary in the country in which it operates or the cash flows in dollars that it sends to its parent...
-
Describe the relationship between a record and a field. How would this look in a database?
-
Jetadiah Brown wants to establish a pet store called Jets Pets. Jet thinks there is an opportunity on the north side of the city, because he knows that many new subdivisions have been built there and...
-
Classify the following items into liabilities and assets: (a) Computers (b) Buildings (c) Trade payables (d) Inventory (e) Trade receivables (f) Cash in bank (g) Bank overdraft (h) Loan from bank (i)...
-
Explain the difference between yield to maturity (YTM) and yield to call (YTC).
-
Herr Corporation has 3,000 shares of 7%, $100 par value preferred stock outstanding at December 31, 2020. At December 31, 2020, the company declared a $105,000 cash dividend. Determine the dividend...
-
Evaluate how many lines there are in a true rotational spectrum of CO molecules whose natural vibration frequency is w = 4.09 1014 s1 and moment of inertia I = 1.44 1039 g cm2.
-
Which of Ne or Ar has the larger van der Waals parameter a? Explain your reasoning.
-
From the following data at 298.15 K as well as data in Table 4.1 (Appendix B, Data Tables), calculate the standard enthalpy of formation of H 2 S(g) and of FeS 2 (s): AR(kJ mol) Fe(s) + 2H2S(g) ...
-
Which of Ne or Ar has the larger van der Waals parameter b? Explain your reasoning.
-
Orion Languages, Incorporated (OLI) offers conversational instruction in several languages. Customers can choose from three approaches: Group, Individual, or Intense. Group customers meet as a part...
-
Payroll is prepared for the month of January. Paychecks will be issued February 7. Annual salary information is as follows: Bryant, $36,000; Lennox, $40,000; Larkin, $26,000. Each employee...
-
Porter Pots ("PP") Pte Ltd manufactures a single product (an earthen self-cooking pot) and uses a standard costing system for control purposes. PP budgeted to produce and sell 14,000 units this year....

Study smarter with the SolutionInn App


