The squares of a number of vibrational energy eigenfunctions are shown superimposed on a Morse potential in
Question:
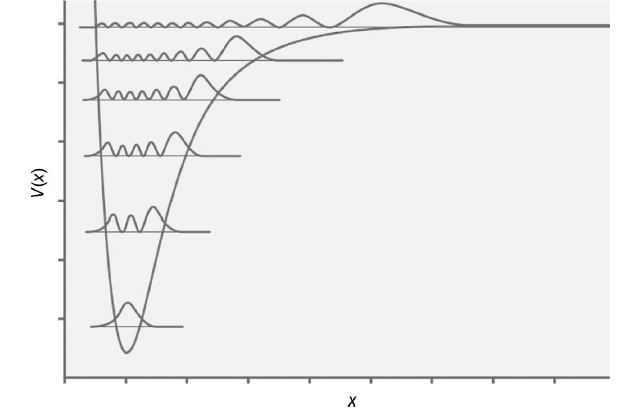
Transcribed Image Text:
franna (x)A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
The quantum number is the number of nodes or 0 2 4 6 8 and 11 from the l...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Overtone transitions in vibrational absorption spectra for which În = +2, + 3, ¦ are forbidden for the harmonic potential V = (1 2)kx 2 because μ mn x = 0 for £m...
-
The total energy eigenvalues for the hydrogen atom are given by E n = e 2 / (8Ïε 0 a 0 n), n = 1, 2, 3, 4,¦, and the three quantum numbers associated with the total energy...
-
In this problem, you will solve for the total energy eigenfunctions and eigenvalues for an electron in a finite depth box. We first go through the calculation for the box parameters used in Figure...
-
A specific purpose statement is not stated in the speech but includes both the general purpose and the topic. It often reveals the intended goal for the audience. O True False
-
In Exercises 1-4, solve the equation. 1. sin 2x sin x = 0 2. sin 2x sin x = cos x 3. cos 2x cos x = 0 4. cos 2x + sin x = 0
-
Min Yachts has accounts receivable of 95,400 at March 31, 2025 (amounts in thousands). Credit terms are 2/10, n/30. At March 31, 2025, there is a 2,100 credit balance in Allowance for Doubtful...
-
Explain the difference between Inverse ETFs and Leverage ETFs.
-
Cost-Cutting Proposals Chatman Machine Shop is considering a four-year project to improve its production efficiency. Buying a new machine press for $530,000 is estimated to result in $205,000 in...
-
A stock just paid a dividend of $1.00. Its dividends are paid annually and are expected to grow 6.00% per year. The stock is trading for $26.50 today. What is the forward dividend yield (D1 / P) on...
-
Twelve different message signals, each with a bandwidth of 10 kHz, are to be multiplexed and transmitted. Determine the minimum bandwidth required for each method if the multiplexing/modulation...
-
The number of molecules in a given energy level is proportional to e E kBT , where E is the difference in energy between the level in question and the ground state. How is it possible that a...
-
As a diatomic molecule rotates, the centrifugal force leads to a small change in the bond length. Do you expect the bond length to increase or decrease? Do you expect the difference between adjacent...
-
Some calculus textbooks define the inverse secant function using the range [0, /2) U [, 3/2). (a) Sketch the graph y = arcsec x using this range. (b) Show that y' 1 -2 xx - 1 -
-
Kamal Corporation is considering purchasing one of two new diagnostic machines. Either machine would make it possible for the company to bid on jobs that it currently isnt equipped to do. Estimates...
-
Pick an organization, such as a local travel agency or supermarket. Describe its organizational domain, then draw a map of the forces in its general and specific environments that affect the way it...
-
Develop a fault tree for events that could lead to release of benzene to the atmosphere for the distillation section designed in problem 9.5. Data from problem 9.5 A distillation column separates...
-
Choose a small organization in your city, such as a restaurant or school, and draw a chart showing its structure. Do you think the number of levels in its hierarchy and the span of control at each...
-
GDM Equipment, Inc. reported the following data for 2016: Compute GDMs net cash provided by operating activitiesindirect method. Income Statement: Net Income Depreciation Expense Balance Sheet:...
-
In problem, solve each equation. e x2 = e 3x 1/e 2
-
1. Following are information about Alhadaf Co. Cost incurred Inventory Purchases Sales Adverting expense Salary Expense Depreciation Beginning Inventory Ending Inventory Amount 118,000 350.000 90,000...
-
A 1.50-L mixture of helium, neon, and argon has a total pressure of 754 mmHg at 310 K. If the partial pressure of helium is 431 mmHg and partial pressure of neon is 211 mmHg, what mass of argon is...
-
Methanol (CH 3 OH) can be synthesized by the reaction: CO(g) + 2 H 2 (g) CH 3 OH(g) What volume (in liters) of methanol gas, measured at a temperature of 473 K and a pressure of 820 mmHg, is...
-
How many grams of water form when 2.41 L of oxygen gas at STP completely react with an appropriate amount of H 2 ? 2 H 2 (g) + O 2 (g) 2 H2O(g)
-
Max Warrie, a famous former Australian cricketer had agreed to play at the Sydney Cricket Ground in an exhibition match against a rest of the world team in the coming Australian summer. He was to be...
-
Does a creditor have an obligation to advise a company to obtain legal advice before signing a contract? In this case, a director of a company is saying they did not know what they were signing when...
-
Randal embezzled $6000 of his employer's money and then disappeared. The employer's accountant called at Randal's residence and found out that his wife had a $3500 term deposit. When the accountant...

Study smarter with the SolutionInn App


