Use the following data to determine the conductance at infinite dilution for NaNO 3 . A, (KCI)
Question:
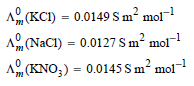
Transcribed Image Text:
A, (KCI) = 0.0149 Sm² mol- A (NaCI) = 0.0127 Sm² mol ² mol- A, (KNO;) = 0.0145 S m
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The above species are strong electrolytes and the ...View the full answer

Answered By

Niala Orodi
I am a competent and an experienced writer with impeccable research and analytical skills. I am capable of producing quality content promptly. My core specialty includes health and medical sciences, but I can competently handle a vast majority of disciplines.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Measurement of diffusivity by unsteady-state evaporation, use the following data to determine the diffusivity of ethyl propionate (species A) into a mixture of 20 mole% air and 80 mole% hydrogen...
-
A furnace fails in a home in the winter. Mercifully, the electric power remains on. The resident engineer (ells her spouse not to worry; they'll move into the kitchen, where the heat discarded from...
-
Use the following data to determine the Langmuir adsorption parameters for nitrogen on mica: V ads (cm 3 g 1 P (Torr) 0.494.2.1 10 3 0.782.4.60 10 3 1.161.30 10 2
-
(1) How Does Strategy Respond to Environmental Factors Imprima Corporation is based in the United States and is examining the prospects for expanding into international markets with its main product,...
-
Describe the error. The graph of a quartic (fourth-degree) polynomial y = f (x) is shown. One of the zeros is i. 10- -20 -30 -40+ The function is f(x) = (x + 2)(x 3.5)(x i). X
-
Figure shows the cross section of a wall made of three layers. The layer thicknesses are L 1 , L 2 = 0.700 L 1 and L 3 = 0.350 L 1 . The thermal conductivities are k 1 , k 2 = 0.900 k 1 and k 3 =...
-
Jennifer is a salesperson for a major chemical company doing business in China. To close a multimillion-dollar contract, she has been asked by a Chinese government official for a payment of...
-
Teri Hatcher Inc., in its first year of operations, has the following differences between the book basis and tax basis of its assets and liabilities at the end of 2008. It is estimated that the...
-
Step 2: Smooth Sailing 1. Edit the sshd_config file. [Your bash commands here]
-
An analysis of the transactions made by S. Moses & Co., a certified public accounting firm, for the month of August is shown below. The expenses were $650 for rent, $4,900 for salaries, and $500 for...
-
A current of 2.00 A is applied to a metal wire for 30.0 s. How many electrons pass through a given point in the wire during this time?
-
The following molar conductivity data are obtained for an electrolyte: Concentration (M) m (Sm 0 mol 1 ) 0.0005..0.01245 0.001.0.01237 0.005.0.01207 0.010.01185 0.02.0.01158 0.05.0.01111 0.1.0.01067...
-
Journalize the following labor- related transactions for Creative Crystal, Ltd. at its plant in Portland, Oregon. Plant janitors wages ......... $ 570 Plant furnace operators wages ..... 880 Glass...
-
If an organization has an average daily gross patient service revenue $230,000 along with 120 patients preregistered encounters, 150 scheduled encounters, and $100,000 in gross dollars in discharged,...
-
XYZ issued 2,000 shares of $8 par value stock to pay for legal services provided by 123 Company. Market Value of the stock was $10 per share. The journal entry will include: a. $16,000 debit to...
-
What strategies can e-commerce sites use to assure customers that they are legitimate and trustworthy? Write in about 150-200 words count
-
If $400,000 will purchase a 20-year annuity paying $930 at the end of each month, what monthly compounded nominal rate and effective rate of interest will the invested funds earn?
-
A $500,000, ten year, 7% bond issue was sold to yield 6% interest payable annually. Actuarial information for 10 periods is as follows: present value $1 @ 7% = 0.50835 @ 6% = 0.55839 present value of...
-
Making use of the Fresnel Equations, show that t || (θ p )t' p|| (θ' p ) = 1, as in the previous problem. Data from Prob. 4.100 A wave, linearly polarized in the...
-
The rate at which the temperature of an object changes is proportional to the difference between its own temperature and the temperature of the surrounding medium. Express this rate as a function of...
-
Use the result of Problem P3.10 to derive a formula for (CV /V ) T for a gas that obeys the RedlichKwong equation of state, RT 1 a Vm - b VT VVm + b)' T VVm P:
-
A 22.0 g mass of ice at 273 K is added to 136 g of H 2 O(l) at 310 K at constant pressure. Is the final state of the system ice or liquid water? Calculate S for the process. Is the process...
-
The function f (x, y) is given by f (x, y) = xy sin 5x + x 2 y in y +3e -2x2 cos y. Determine and Obtain an expression for the total differential df. 2 f af a (af ay ax of ay? ax ay a (af a (af ...
-
The controller for Tulsa Medical Supply Company has established the following activity cost pools and cost drivers. Machine setups Budgeted Overhead Cost Cost Driver Number of setups Weight of raw...
-
In 2023, Miranda records net earnings from self-employment of $168,500. She has no other income. Determine the amount of Miranda's self-employment tax and her AGI income tax deduction. In your...
-
:48 t int A company had the following assets and liabilities at the beginning and end of this year. Beginning of the year End of the year Assets $ 82,500 135,500 Liabilities $ 35,368 54,878 a. Owner...

Study smarter with the SolutionInn App


