What is the functional dependence of the 1s orbital energy on Z in Figure 21.7? Check your
Question:
Figure 21.7
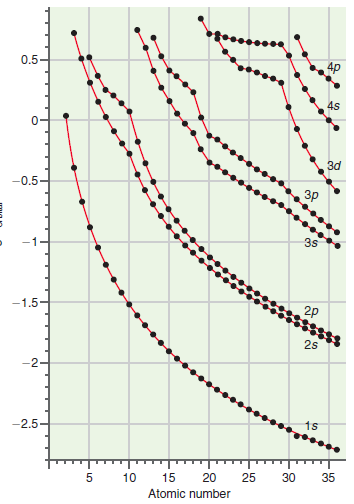
Transcribed Image Text:
0.5 4s Зd -0.5 зр 3s 2p -1.5 2s 15 -2.5 35 30 25 20 10 Atomic number -15
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
The radial equation for a one electron atom with nuclear charge Z ...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Electron and hole concentrations increase with temperature. For pure silicon, suitable expressions are h = e = 6200T 1.5 e 7000 /T C/m 3 . The functional dependence of the mobilities on temperature...
-
Extend the considerations of the preceding problem to particle diffusion, and assume that there is a net particle generation rate gu that is proportional to the local particle concentration, gu =...
-
Vortex flow (a) Show that the complex potential w = (iT/2) in z describes the flow in a vortex. Verify that the tangential velocity is given by v = T/2 and that v r = 0. This type of flow is...
-
Brennan Physiotherapies had a beta of 0.85. Reasonable estimates for the RF and the required rate of return on the market, R(Rm) were 7% and 15%, respectively. What is the required rate of return on...
-
A privately owned yacht leaves a dock in Myrtle Beach, South Carolina, and heads toward Freeport in the Bahamas at a bearing of S 1.4 E. The yacht averages a speed of 20 knots over the...
-
This case is based on an actual situation. Centennial Construction Company, headquartered in Dallas, Texas, built a Rodeway Motel 35 miles north of Dallas. The construction foreman, whose name was...
-
(a) In Figure 7.5, what is the momentum of the ball during the collision? (b) Is the momentum of the ball constant before, during, and after the collision? If so, why? If not, why not, and for what...
-
York Industries leases a large specialized machine to Echo Company at a total rental of $1,400,000, payable in five annual installments in the following declining pattern: 24% in the first two years,...
-
what happens when ou use an aggregation function in a calculated column?
-
Steve Austin is the fleet manager for SharePlane, a company that sells fractional ownership of private jets. SharePlane must carefully maintain their jets at all times. If a jet breaks down, it must...
-
The total energy eigenvalues for the hydrogen atom are given by E n = e 2 / (8Ïε 0 a 0 n), n = 1, 2, 3, 4,¦, and the three quantum numbers associated with the total energy...
-
Why is the total energy of a many-electron atom not equal to the sum of the orbital energies for each electron?
-
In Problems 4148, find an equation for the hyperbola described. Graph the equation. Vertices at (-1,-1) and (3,-1); 3 asymptote the line y + 1 = 2 (x - 1)
-
Alpha Company is projecting its sales for the next three months and has generated the following inputs from its sales manager. April May June Units sold 5,000 5,300 4,800 Unit sales price $10.00...
-
The defective copper tubing from Transaction 28 is returned to Edward's Plumbing Supplies, Inc. along with a debit memo in the amount of $3,744 against future purchases with Edward's Plumbing...
-
Received an invoice from DeKalb Transport for $2,300 for freight costs (on sales shipments) incurred during the past 30 days, terms n/30. Freight costs are charged to delivery expense when incurred....
-
Gearhead started out selling bicycles, but later added shoes, and then many other outdoor activity related items. this is an example of how gearhead's product assortment has multiple choice gotten...
-
The accounting practices of not-for-profits are more compatible with those of business than with those of government. Like governments, not-for-profits account for their resources in funds. How funds...
-
In each of the following situations, briefly explain whether the IS curve will shift and, if it does shift, in which direction it will shift: a. Consumers become more optimistic about their future...
-
X-1 Find the domain of the function f(x) : x 1 2 - O (-00, -1) U (-1, ) O (-00, 1) U (1, ) O -00, -1) U (-1, 1) U (1, 0) O (- 1, 1)
-
Calculate the percentage change in K x for the reaction H 2 CO(g) CO(g) + H 2 (g) when the total pressure is increased from 1.0 bar to 2.0 bar at constant temperature.
-
A sealed container was filled with 0.300 mol H 2 (g), 0.400 mol I 2 (g), and 0.200 mol HI(g) at 870 K and total pressure 1.00 bar. Calculate the amounts of the components in the mixture at...
-
The equilibrium constant for the gas-phase isomerization of borneol (C 10 H 17 OH) to isoborneol at 503 K is 0.106. A mixture consisting of 7.50 g of borneol and 14.0 g of isoborneol in a container...
-
a) Give examples of low, medium and high strain rate test types. [4 marks] b) A plot of log (stress) against log (strain rate) provides a straight line relationship. Based on this, provide an...
-
3- Let the density of the pyramid material be p = 2000 kg/m. Determine the average normal stress at a cross section located at x measured from the apex. Express the stress as a function of x. Assume...
-
You are designing a wing with no aerodynamic twist based on the airfoil camber given by: N C 3.1 (H-H) = 0.1 The wing will have an aspect ratio of 10, a taper ratio of 0.8, and a constant geometric...

Study smarter with the SolutionInn App


